The mechanism of bromination of an alkene, which involves a cyclic bromonium ion, explains why trans-2-butene and cis-2-butene give rise, respectively, to a meso structure and a racemic mixture of 2,3-dibromobutanes Me Bra Br trans-but-2-ene Meso product Brz Me Me Me Me cis-but-2-ene Racemic product -....in ior 4. Make a model and draw the structure of the cyclic bromonium ion that results from addition to cis-2-butene 5. Make a model of the structure of the racemic products that result from opening of this bromonium ion by an SN2 mechanism 6. For each enantiomeric structure assign the configuration at each centre
The mechanism of bromination of an alkene, which involves a cyclic bromonium ion, explains why trans-2-butene and cis-2-butene give rise, respectively, to a meso structure and a racemic mixture of 2,3-dibromobutanes Me Bra Br trans-but-2-ene Meso product Brz Me Me Me Me cis-but-2-ene Racemic product -....in ior 4. Make a model and draw the structure of the cyclic bromonium ion that results from addition to cis-2-butene 5. Make a model of the structure of the racemic products that result from opening of this bromonium ion by an SN2 mechanism 6. For each enantiomeric structure assign the configuration at each centre
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 36AP
Related questions
Question
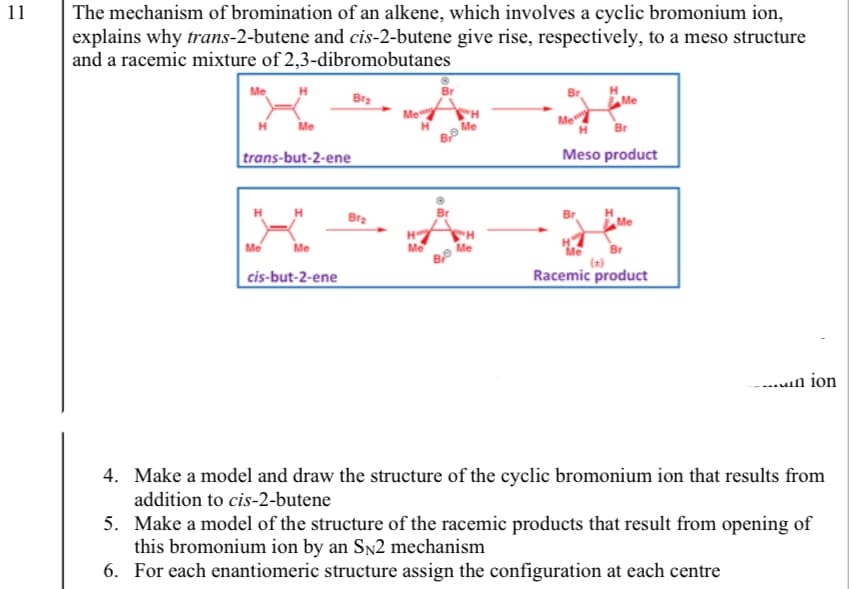
Transcribed Image Text:11
The mechanism of bromination of an alkene, which involves a cyclic bromonium ion,
explains why trans-2-butene and cis-2-butene give rise, respectively, to a meso structure
and a racemic mixture of 2,3-dibromobutanes
Me
Bra
Me
H
trans-but-2-ene
Meso product
H
H
Bra
Me
Me
Me
Me
Me
Br
cis-but-2-ene
Racemic product
-..un ion
4. Make a model and draw the structure of the cyclic bromonium ion that results from
addition to cis-2-butene
5. Make a model of the structure of the racemic products that result from opening of
this bromonium ion by an Sn2 mechanism
6. For each enantiomeric structure assign the configuration at each centre
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

