The mermaid need to go talk to fairies about the water contamination in the kingdom of fuzzy blue unicorns. The fairies live very high up in elevation, this creates a problem for the mermaids who can walk but do not climb mountains well. In order for the mermaid to meet with the fairies they have constructed hot air balloons. The mermaid (keep in mind mermaid are very small) have found they can generate hot carbon dioxide from the reaction of elemental carbon and ozone. If the balloon has a volume of 3.571 L at 16.93 °C and 597.0 torr, how many grams of carbon is needed to fill the balloon complete with carbon dioxide? (Enter your final numerical answer here with 3 decimal places and no units, the understood unit is grams) C(6) + O3(9) CO2%9)
The mermaid need to go talk to fairies about the water contamination in the kingdom of fuzzy blue unicorns. The fairies live very high up in elevation, this creates a problem for the mermaids who can walk but do not climb mountains well. In order for the mermaid to meet with the fairies they have constructed hot air balloons. The mermaid (keep in mind mermaid are very small) have found they can generate hot carbon dioxide from the reaction of elemental carbon and ozone. If the balloon has a volume of 3.571 L at 16.93 °C and 597.0 torr, how many grams of carbon is needed to fill the balloon complete with carbon dioxide? (Enter your final numerical answer here with 3 decimal places and no units, the understood unit is grams) C(6) + O3(9) CO2%9)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter10: Solids, Liquids, And Phase Transitions
Section: Chapter Questions
Problem 56AP
Related questions
Question
100%
Please explain properly and neatly
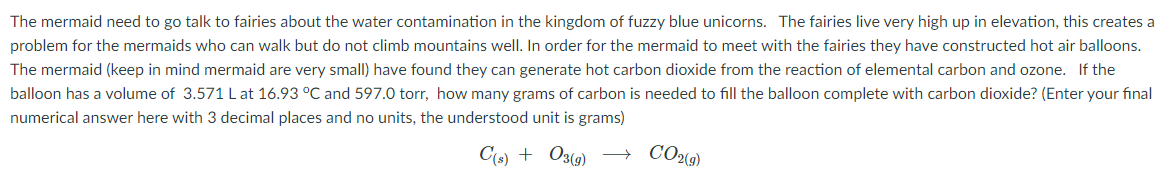
Transcribed Image Text:The mermaid need to go talk to fairies about the water contamination in the kingdom of fuzzy blue unicorns. The fairies live very high up in elevation, this creates a
problem for the mermaids who can walk but do not climb mountains well. In order for the mermaid to meet with the fairies they have constructed hot air balloons.
The mermaid (keep in mind mermaid are very small) have found they can generate hot carbon dioxide from the reaction of elemental carbon and ozone. If the
balloon has a volume of 3.571 L at 16.93 °C and 597.0 torr, how many grams of carbon is needed to fill the balloon complete with carbon dioxide? (Enter your final
numerical answer here with 3 decimal places and no units, the understood unit is grams)
C(6) + O3(9)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning