The pka of formaldehyde (H2C=0) is not listed in Table 6-1. (a) Based on your understanding of charge stability, which of the following compounds (A-F) would you expect to have a pKa most similar to that of formaldehyde? C. H H3C-OH H3C-CH3 H,C=CH2 HC=CH H-C=N HO. A В E F (b) Using Table 6-1, as well as your knowledge of the factors that affect charge stability, estimate the pka of formaldehyde. TABLE 61 Values of Ka and pka for Various Acids Acid Conjugate Base Ka pK. Acid Conjugate Base Ka pKa CH,OH Methanol CH,00 3.2 x 1014 15.5 F,C- 1x 10 -13 2 к 10-48 15.7 Trifluoromethanesulfonic acid Water HO Ethanol 1 к 1048 16 HO-S-OH 1x 10° -9 Sulfuric acid -OH 3.2 x 10-7 16.5 HCI 1 к 107 -7 Propan 2-ol (Isopropyl alcohol) Hydrochloric acid 55 -1.7 Hydronium lon OH 1x 10-10 19 Methylpropan-2-ol (tert-Butyl alcohol) 0.17 0.77 ClC Trichloroethanoic acid (Trichloroacetic acid) 1x 10-20 20 FO 6.3 x 10 CH, Propanone (Acetone) HF 3.2 Hydrofluoric acid HC=CH HCECO 1x 10 25 Ethyne (Acetylene) 6.3 x 10 4.2 NH, 1x 107 27 Benzolc acid Aniline (Phenylamine) 1.8 x 10 4.75 1 к 10- 35 он Ethanoic acid (Acetic acid) Hydrogen gas HSO 6.3 x 10 7.2 1x 10- 38 CH, H.C N-Methylmethanamine (Dimethylamine) Hydrogen sulfide CH, HNO NH, 4x 10-0 9.4 Ammonium ion 1 х 10-44 44 H,C=CH, Ethene (Ethylene) 1x 10-10 10.0 Ho -1 x 10-4 -45 Phenol Ethoxyethane H,C-NH, Methylammonium ion H,C-NH, 2.3 x 101 10.63 (Diethyl ether) CH4 1х 10 4а 48 Methane HO. 4 x 10-1 12.4 2,22-Trifluoroethanol 1x 10 t0 CH,CH, Ethane 50 5.0 x 10- 14.3 2-Chloroethanol pk,--log K The lesa positive (or more negative the pk, value, the stranger the acid relative to inother acid.
The pka of formaldehyde (H2C=0) is not listed in Table 6-1. (a) Based on your understanding of charge stability, which of the following compounds (A-F) would you expect to have a pKa most similar to that of formaldehyde? C. H H3C-OH H3C-CH3 H,C=CH2 HC=CH H-C=N HO. A В E F (b) Using Table 6-1, as well as your knowledge of the factors that affect charge stability, estimate the pka of formaldehyde. TABLE 61 Values of Ka and pka for Various Acids Acid Conjugate Base Ka pK. Acid Conjugate Base Ka pKa CH,OH Methanol CH,00 3.2 x 1014 15.5 F,C- 1x 10 -13 2 к 10-48 15.7 Trifluoromethanesulfonic acid Water HO Ethanol 1 к 1048 16 HO-S-OH 1x 10° -9 Sulfuric acid -OH 3.2 x 10-7 16.5 HCI 1 к 107 -7 Propan 2-ol (Isopropyl alcohol) Hydrochloric acid 55 -1.7 Hydronium lon OH 1x 10-10 19 Methylpropan-2-ol (tert-Butyl alcohol) 0.17 0.77 ClC Trichloroethanoic acid (Trichloroacetic acid) 1x 10-20 20 FO 6.3 x 10 CH, Propanone (Acetone) HF 3.2 Hydrofluoric acid HC=CH HCECO 1x 10 25 Ethyne (Acetylene) 6.3 x 10 4.2 NH, 1x 107 27 Benzolc acid Aniline (Phenylamine) 1.8 x 10 4.75 1 к 10- 35 он Ethanoic acid (Acetic acid) Hydrogen gas HSO 6.3 x 10 7.2 1x 10- 38 CH, H.C N-Methylmethanamine (Dimethylamine) Hydrogen sulfide CH, HNO NH, 4x 10-0 9.4 Ammonium ion 1 х 10-44 44 H,C=CH, Ethene (Ethylene) 1x 10-10 10.0 Ho -1 x 10-4 -45 Phenol Ethoxyethane H,C-NH, Methylammonium ion H,C-NH, 2.3 x 101 10.63 (Diethyl ether) CH4 1х 10 4а 48 Methane HO. 4 x 10-1 12.4 2,22-Trifluoroethanol 1x 10 t0 CH,CH, Ethane 50 5.0 x 10- 14.3 2-Chloroethanol pk,--log K The lesa positive (or more negative the pk, value, the stranger the acid relative to inother acid.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter8: Addition Via Carbocation
Section: Chapter Questions
Problem 35CTQ
Related questions
Question
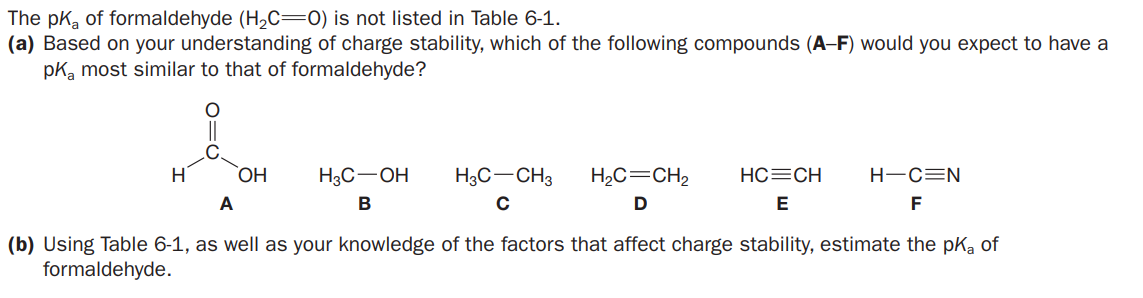
Transcribed Image Text:The pka of formaldehyde (H2C=0) is not listed in Table 6-1.
(a) Based on your understanding of charge stability, which of the following compounds (A-F) would you expect to have a
pKa most similar to that of formaldehyde?
C.
H
H3C-OH
H3C-CH3
H,C=CH2
HC=CH
H-C=N
HO.
A
В
E
F
(b) Using Table 6-1, as well as your knowledge of the factors that affect charge stability, estimate the pka of
formaldehyde.

Transcribed Image Text:TABLE 61 Values of Ka and pka for Various Acids
Acid
Conjugate Base
Ka
pK.
Acid
Conjugate Base
Ka
pKa
CH,OH
Methanol
CH,00
3.2 x 1014 15.5
F,C-
1x 10 -13
2 к 10-48
15.7
Trifluoromethanesulfonic acid
Water
HO
Ethanol
1 к 1048
16
HO-S-OH
1x 10°
-9
Sulfuric acid
-OH
3.2 x 10-7
16.5
HCI
1 к 107
-7
Propan 2-ol
(Isopropyl alcohol)
Hydrochloric acid
55
-1.7
Hydronium lon
OH
1x 10-10
19
Methylpropan-2-ol
(tert-Butyl alcohol)
0.17
0.77
ClC
Trichloroethanoic acid
(Trichloroacetic acid)
1x 10-20
20
FO
6.3 x 10
CH,
Propanone (Acetone)
HF
3.2
Hydrofluoric acid
HC=CH
HCECO
1x 10
25
Ethyne (Acetylene)
6.3 x 10
4.2
NH,
1x 107
27
Benzolc acid
Aniline (Phenylamine)
1.8 x 10
4.75
1 к 10-
35
он
Ethanoic acid (Acetic acid)
Hydrogen gas
HSO
6.3 x 10
7.2
1x 10-
38
CH,
H.C
N-Methylmethanamine
(Dimethylamine)
Hydrogen sulfide
CH,
HNO
NH,
4x 10-0
9.4
Ammonium ion
1 х 10-44
44
H,C=CH,
Ethene (Ethylene)
1x 10-10
10.0
Ho
-1 x 10-4 -45
Phenol
Ethoxyethane
H,C-NH,
Methylammonium ion
H,C-NH,
2.3 x 101 10.63
(Diethyl ether)
CH4
1х 10 4а
48
Methane
HO.
4 x 10-1
12.4
2,22-Trifluoroethanol
1x 10 t0
CH,CH,
Ethane
50
5.0 x 10- 14.3
2-Chloroethanol
pk,--log K The lesa positive (or more negative the pk, value, the stranger the acid relative to inother acid.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 7 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
