The plot below from your book does NOT have data for ammonia. 1.0 72.00 7-1.50 0.8 71.20 0.6 z. T-1.19, 0.4 • Nitroges Methane a Ethane Ethylene • Propane -Butane Isopentane -Heptane . Carbon dioxide Water 0.2 T-1.00 0.0 4. PR However, you can use this plot (an example of the law of corresponding states) to calculate the molar volume (V) of ammonia at a temperature and pressure of interest. What information do you need in order to do this? O The critical temperature and pressure for ammonia. O The standard molar Gibbs energy for ammonia. O The normal boiling point for ammonia. O The vibrational spacing in ammonia.
The plot below from your book does NOT have data for ammonia. 1.0 72.00 7-1.50 0.8 71.20 0.6 z. T-1.19, 0.4 • Nitroges Methane a Ethane Ethylene • Propane -Butane Isopentane -Heptane . Carbon dioxide Water 0.2 T-1.00 0.0 4. PR However, you can use this plot (an example of the law of corresponding states) to calculate the molar volume (V) of ammonia at a temperature and pressure of interest. What information do you need in order to do this? O The critical temperature and pressure for ammonia. O The standard molar Gibbs energy for ammonia. O The normal boiling point for ammonia. O The vibrational spacing in ammonia.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter3: Measurement And Chemical Calculations
Section: Chapter Questions
Problem 99E
Related questions
Question
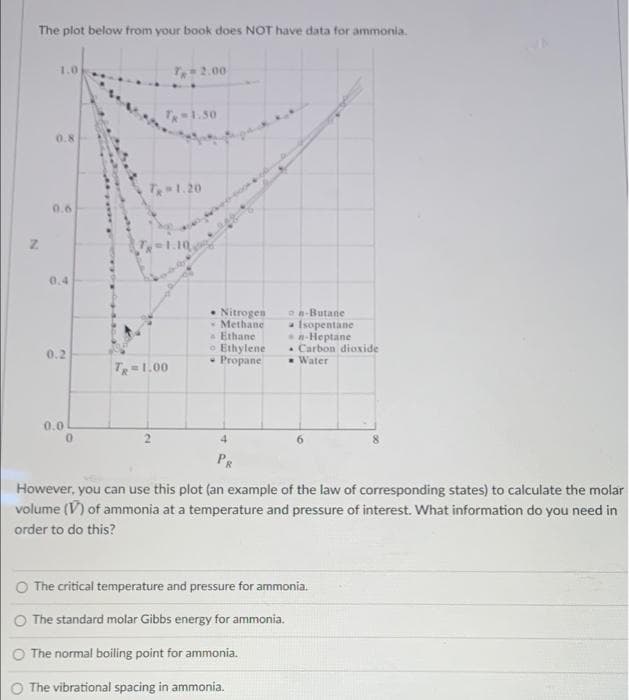
Transcribed Image Text:The plot below from your book does NOT have data for ammonia.
1.0
T2.00
T1.50
0.8
T-1.20
0.6
T1.10
0.4
• Nitrogen
* Methane
a Ethane
o Ethylene
- Propane
an-Butane
- Isopentane
n-Heptane
. Carbon dioxide
*Water
0.2
T=1.00
0.0
4.
PR
However, you can use this plot (an example of the law of corresponding states) to calculate the molar
volume (V) of ammonia at a temperature and pressure of interest. What information do you need in
order to do this?
O The critical temperature and pressure for ammonia.
The standard molar Gibbs energy for ammonia.
O The normal boiling point for ammonia.
O The vibrational spacing in ammonia.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
