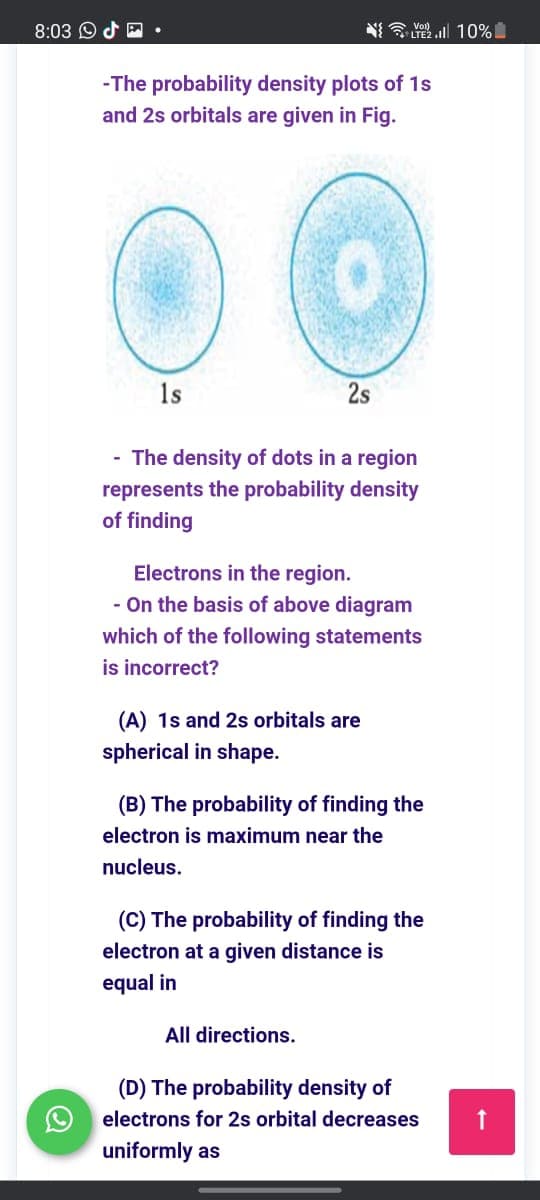
-The probability density plots of 1s and 2s orbitals are given in Fig. 1s 2s - The density of dots in a region represents the probability density of finding Electrons in the region. - On the basis of above diagram which of the following statements is incorrect? (A) 1s and 2s orbitals are spherical in shape. (B) The probability of finding the electron is maximum near the nucleus. (C) The probability of finding the electron at a given distance is equal in All directions. (D) The probability density of O electrons for 2s orbital decreases uniformly as
-The probability density plots of 1s and 2s orbitals are given in Fig. 1s 2s - The density of dots in a region represents the probability density of finding Electrons in the region. - On the basis of above diagram which of the following statements is incorrect? (A) 1s and 2s orbitals are spherical in shape. (B) The probability of finding the electron is maximum near the nucleus. (C) The probability of finding the electron at a given distance is equal in All directions. (D) The probability density of O electrons for 2s orbital decreases uniformly as
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter5: Quantum Mechanics And Atomic Structure
Section: Chapter Questions
Problem 6P: Using Table 5.2, write down the mathematical expression for the 2px wave function for an...
Related questions
Question
Transcribed Image Text:8:03 O d
N E Yoll 10%
LTE2
-The probability density plots of 1s
and 2s orbitals are given in Fig.
1s
2s
- The density of dots in a region
represents the probability density
of finding
Electrons in the region.
- On the basis of above diagram
which of the following statements
is incorrect?
(A) 1s and 2s orbitals are
spherical in shape.
(B) The probability of finding the
electron is maximum near the
nucleus.
(C) The probability of finding the
electron at a given distance is
equal in
All directions.
(D) The probability density of
electrons for 2s orbital decreases
uniformly as
Transcribed Image Text:8:37
N Yll 5%
+LTE2
- What does spectroscope do?
(A) Collect light to identify element
(B) Refract light
(C) Absorb light
(D) Transmit light
a
b
d
Previous
Next
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,