The relative polarities of a few common solvents are given below: Solvent name Relative polarity Water 1 Acetic acid 0.65 Acetonitrile 0.48 While using a C-18 hydrocarbon column for protein purification, which of the following will be the best elution solvent mixture? The final polarity of the solvent mixture can be predicted by considering the relative % of each constituent and their respective relative polarity values provide. 1. 90% water and 10% acetic acid II. 90% acetic acid and 10% acetonitrile III. 90% acetonitrile and 10% water IV. 10% acetonitrile and 90% acetic acid
The relative polarities of a few common solvents are given below: Solvent name Relative polarity Water 1 Acetic acid 0.65 Acetonitrile 0.48 While using a C-18 hydrocarbon column for protein purification, which of the following will be the best elution solvent mixture? The final polarity of the solvent mixture can be predicted by considering the relative % of each constituent and their respective relative polarity values provide. 1. 90% water and 10% acetic acid II. 90% acetic acid and 10% acetonitrile III. 90% acetonitrile and 10% water IV. 10% acetonitrile and 90% acetic acid
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter10: Fuels, Organic Chemicals, And Polymers
Section: Chapter Questions
Problem 119QRT
Related questions
Question
Aa.17.
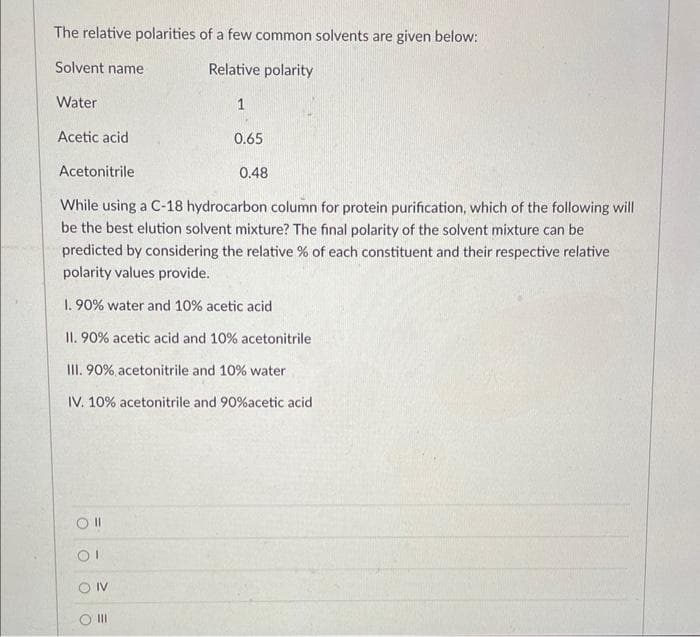
Transcribed Image Text:The relative polarities of a few common solvents are given below:
Relative polarity
Solvent name
Water
Acetic acid
Acetonitrile
CO II
Ol
IV
1
While using a C-18 hydrocarbon column for protein purification, which of the following will
be the best elution solvent mixture? The final polarity of the solvent mixture can be
predicted by considering the relative % of each constituent and their respective relative
polarity values provide.
1. 90% water and 10% acetic acid
II. 90% acetic acid and 10% acetonitrile
III. 90% acetonitrile and 10% water
IV. 10% acetonitrile and 90% acetic acid
|||
0.65
0.48
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole