The structures of six organic compounds are shown below. `NH2 HO OH (b) (a) (c) HO (d) (e) (f) (a) Give the name(s) of the functional group(s) in each compound. All compounds have at least one onal group; some hav e more than one. (b) Give the letter(s) of the compound(s) that are aromatic. Write "None" if there are no aromatic punds. (c) Give the letter(s) of the compound(s) that have optical isomers. Write "None" if no compound has l isomers. (d) What is the molecular formula of (d) ? (e) What is the molecular formula of (c)
The structures of six organic compounds are shown below. `NH2 HO OH (b) (a) (c) HO (d) (e) (f) (a) Give the name(s) of the functional group(s) in each compound. All compounds have at least one onal group; some hav e more than one. (b) Give the letter(s) of the compound(s) that are aromatic. Write "None" if there are no aromatic punds. (c) Give the letter(s) of the compound(s) that have optical isomers. Write "None" if no compound has l isomers. (d) What is the molecular formula of (d) ? (e) What is the molecular formula of (c)
Chapter3: Organic Compounds: Alkanes And Their Stereochemistry
Section3.SE: Something Extra
Problem 50AP: Formaldehyde, H2C=O, is known to all biologists because of its usefulness as a tissue preservative....
Related questions
Question
Answer a-e
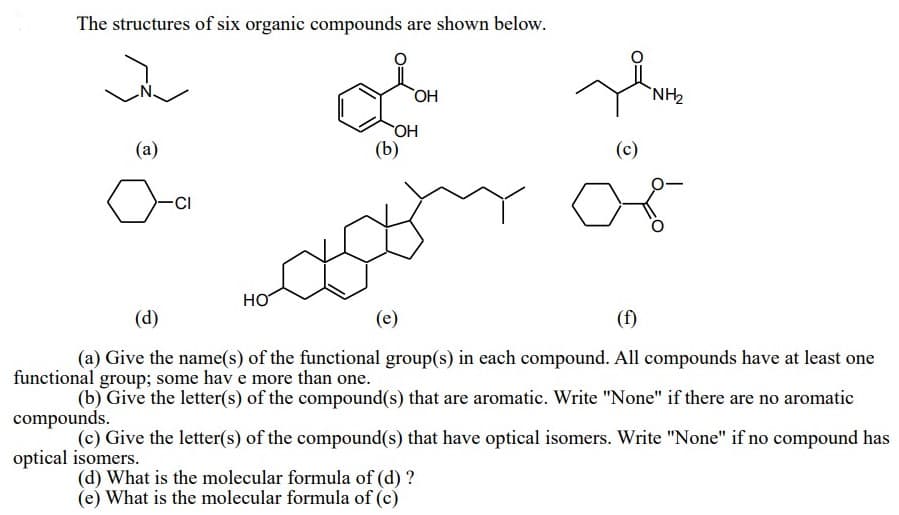
Transcribed Image Text:The structures of six organic compounds are shown below.
ОН
`NH,
(a)
(b)
(c)
CI
HO
(d)
(e)
(f)
(a) Give the name(s) of the functional group(s) in each compound. All compounds have at least one
functional group; some hav e more than one.
(b) Give the letter(s) of the compound(s) that are aromatic. Write "None" if there are no aromatic
compounds.
(c) Give the letter(s) of the compound(s) that have optical isomers. Write "None" if no compound has
optical isomers.
(d) What is the molecular formula of (d) ?
(e) What is the molecular formula of (c)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

