The van der Waals equation of state was designed to predict the relationship between pressure p, volume V and temperature I for gases better than the Ideal Gas Law: p+a |(V-nb) = nRT The van der Waals equation of state. R stands for the gas constant and n for the moles of gas. The van der Waals parameters a and b have to be determined for each gas by comparing to experimental data. Consider the hypothetical gases X, Y and Z listed in the table below. Use the information provided to decide the relative size of the van der Waals parameters a and b. gas boiling point °C molar mass g/mol size of a size of b X -27.4 37. (Choose one) (Choose one)▼ Y 10.7 28. (Choose one) (Choose one) N -6.7 42. (Choose one) (Choose one) 미
The van der Waals equation of state was designed to predict the relationship between pressure p, volume V and temperature I for gases better than the Ideal Gas Law: p+a |(V-nb) = nRT The van der Waals equation of state. R stands for the gas constant and n for the moles of gas. The van der Waals parameters a and b have to be determined for each gas by comparing to experimental data. Consider the hypothetical gases X, Y and Z listed in the table below. Use the information provided to decide the relative size of the van der Waals parameters a and b. gas boiling point °C molar mass g/mol size of a size of b X -27.4 37. (Choose one) (Choose one)▼ Y 10.7 28. (Choose one) (Choose one) N -6.7 42. (Choose one) (Choose one) 미
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter5: The Gaseous State
Section: Chapter Questions
Problem 5.103QP: Calculate the molar volume of ethane at 1.00 atm and 0C and at 10.0 atm and 0C, using the van der...
Related questions
Question
help me
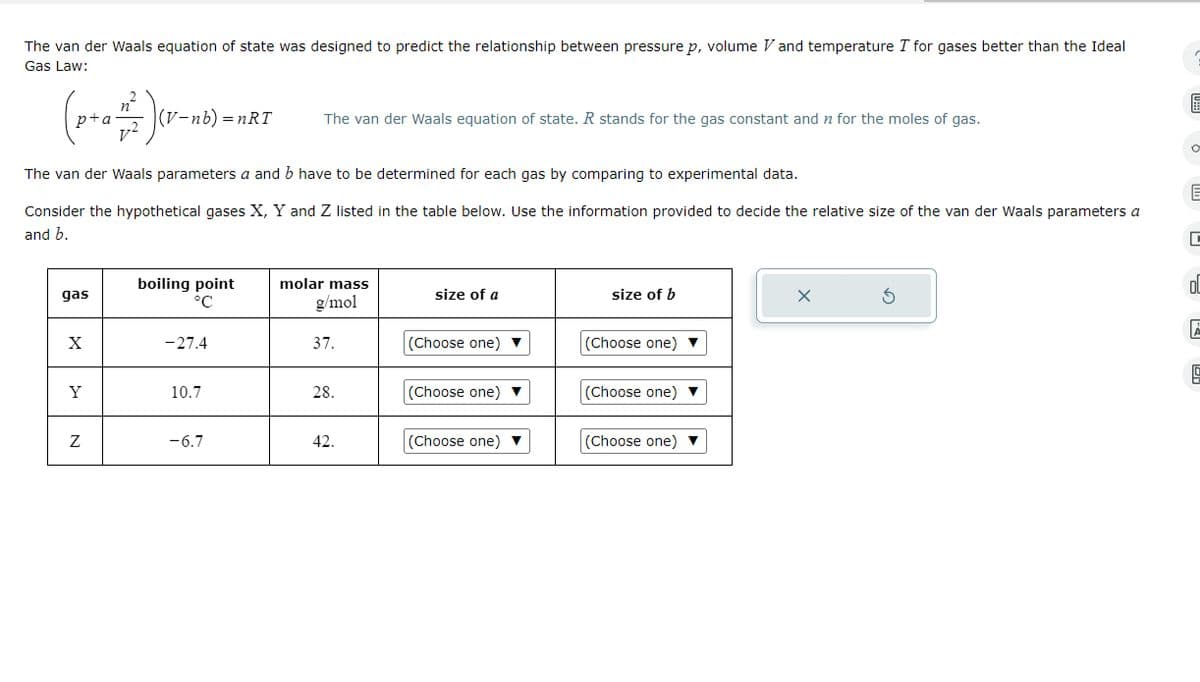
Transcribed Image Text:The van der Waals equation of state was designed to predict the relationship between pressure p, volume V and temperature I for gases better than the Ideal
Gas Law:
p+a
|(V-nb) = nRT
The van der Waals equation of state. R stands for the gas constant and n for the moles of gas.
The van der Waals parameters a and b have to be determined for each gas by comparing to experimental data.
Consider the hypothetical gases X, Y and Z listed in the table below. Use the information provided to decide the relative size of the van der Waals parameters a
and b.
gas
boiling point
°C
molar mass
g/mol
size of a
size of b
X
-27.4
37.
(Choose one)
(Choose one)▼
Y
10.7
28.
(Choose one)
(Choose one)
N
-6.7
42.
(Choose one)
(Choose one)
미
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning
