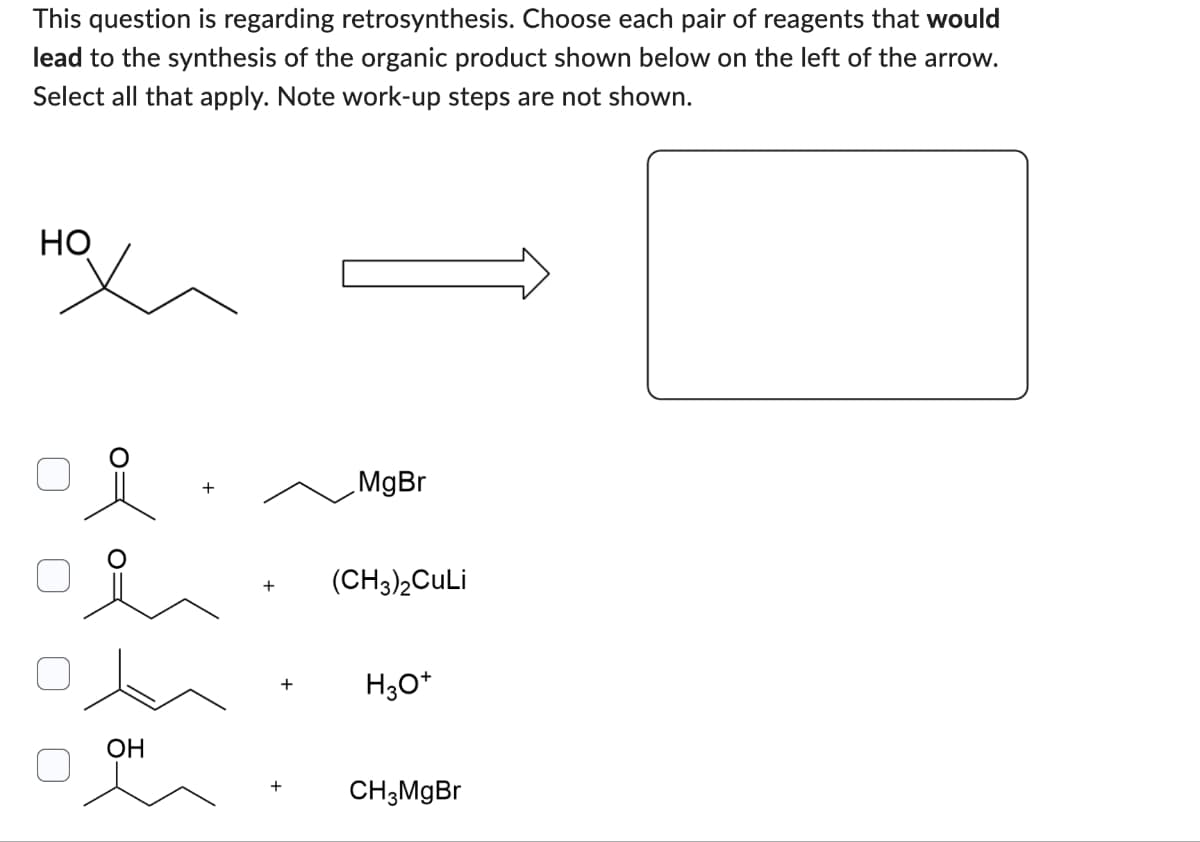
This question is regarding retrosynthesis. Choose each pair of reagents that would lead to the synthesis of the organic product shown below on the left of the arrow. Select all that apply. Note work-up steps are not shown. нох + MgBr (CH3)2CuLi H3O+ OH + CH3MgBr
Q: Consider the leaching of CuO at 28°C. Using the pH of the solution taken at different periods of…
A: Given, Arrhenius constant is 80000“Since you have posted a question with multiple sub parts, we will…
Q: Fix this for me
A: Step 1: Step 2: Step 3: Step 4:
Q: о enzyme + O. о O NH3 Classify the enzyme based on its reaction O NH3 + Oxidoreductase Transferase…
A: The objective of this question is to classify which type of enzyme is used in a given…
Q: Strontium-90 is radioactive and has a half life of 28.8 years. Calculate the activity of a 4.0 mg…
A: The objective of this question is to calculate the activity of a 4.0 mg sample of strontium-90, a…
Q: The hydronium ion concentration in an aqueous solution at 25°C is 5.8×10-3 M.The hydroxide ion…
A: The objective of this question is to find the pH, pOH and hydroxide ion concentration of an aqueous…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 0 RCO H Drawing I 1 I
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: An unknown white powder have been seized by the police on the country border. The chemical analysis…
A: The objective of the given question is propose analytical strategy to establish whether the powder…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Choose the best reagents to complete the following reaction. Br2 A excess Br2, NaOH B 0 CH3CO2H D OH…
A: When methyl ketone is treated with excess halogen and base a reaction occurs leading to the…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Solution 1: 1.3 mol NaOH added to 1.0 L of 1.3 M HCH3CO₂ acids: CH3COOH . It is a weak acid, and…
Q: 1) LDA, 0°C 2) Br b) 1) LDA, -78°C 2) Br 2 D DIBALH, -78°C H₂O 2) 3) NaOH(aq)
A: LDA i.e. Lithium diisopropylamide is a strong base used in the deprotonation reaction of less acidic…
Q: None
A: A. Mo(CO)5PR3 This molecule belongs to the C3v issue institution. The C-O stretching modes will…
Q: An a(1 - 4) glycosidic bond is found in which of the following? O all of these снон H Сон H H OH он…
A:
Q: CH3 NH-C-CH CH3 QUESTION 4 Which of the following molecules is the most selective ẞ1 antagonist…
A: Beta blockers, sometimes written as β-blockers, are a family of drugs that are primarily used to…
Q: Calculate the pH for each of the cases in the titration of 35.0 mL of 0.130 M KOH(aq) with 0.130 M…
A: Answer:In this question calculate the pH of three types of solutions:1. Base solution2. Salt…
Q: i Provide the missing curved arrow notation. ס12 + од H C N O S F P C1 Br
A:
Q: Task: For the reactions below, decide whether the nucleophilic aromatic substitution shown proceeds…
A: When the leaving group is substituted by the nucleophile, the reaction is called nucleophilic…
Q: e: Propose a synthesis prepare: from: from 1° R-X H (Ph-CH₂-X) HO from 1° R-X (CH3CH2-X) Ph H3CO…
A: In this reaction, First, we add a base (NaOEt) in malonic ester which abstract a hydrogen from it…
Q: Iodine, I,, is a solid at room temperature but sublimes (converts from a solid into a gas) when…
A: If you have any questions let me know in comment box thankyou for your question.
Q: The common substrate for all adrenal steroids is: Question 17 options:…
A: The question is asking for the common substrate used in the production of all adrenal steroids.…
Q: If NaOH is combined with CH3OCH 3, which of the following reaction diagrams best represents the…
A: The question is asking us to determine the reaction diagram that best represents the reaction…
Q: der these chemical species by increasing pH of an 0.1 M aqueous solution of each. That is, imagine…
A: All has been ranked higher to lower ph.. H2O. 4.HN3-. 3C5H5N-. 7F-. 6.N3-. 5OH-. 8C5H5NH+. 3HF-.…
Q: Question 10 A photon is quantum of electromagnetic radiation. A True B False
A: The objective of the question is to verify the statement that a photon is a quantum of…
Q: sample size
A: The resulting polymer will be a random copolymer with a blocky characteristic. Chemical composition…
Q: NH 1. NaOH 2. H₂C. CH3 CI Оме ? 3. NaOH, A
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: According to the following graph, the activity of the enzyme decreases more sharply at high…
A: I. As temperature is increased, the substrate binding slowly becomes poorer. This is true. At high…
Q: Show how you would synthesize the following compound using 2-methylhex-1-ene starting materials and…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. Convert ethyne to 2,3-pentanedione (a compound responsible for the aroma of cereal coffee brew).…
A: step 1:Alkylation of terminal alkyne (Or alkylation of acetylide anion):- Terminal Alkynes (having…
Q: 4. Draw the reaction for the chlorination of 2,4,5-trimethyloctane. (May have multiple products)
A: The objective of this question is to draw the reaction for the chlorination of .The reaction in…
Q: What ratio of NaCN to HCN is needed to prepare a pH 9.90 buffer? (Ka of HCN is 4.9 × 10⁻¹⁰)
A: The objective of this question is to find the ratio of sodium cyanide (NaCN) to hydrogen cyanide…
Q: stion 3 yChart - Sch... Home - Profession... Enroll Enr 15 pts To preview image click here For the…
A: Step 1: resonsnce contributors should always have same formal charge and no of bonds only A and F…
Q: Write the equillibrium expression for C(s) + 2N2O(g) CO2(g) + 2N2(g)
A: The objective of the question is to write the equilibrium expression for the given chemical…
Q: Predict the products of this organic reaction: + H₂O A Specifically, in the drawing area below draw…
A: Given reactant:So, the compound has an ester group.
Q: ← Draw the products of the reaction shown below. Ignore inorganic byproducts. CH3(O)CI AICI 3…
A: This is a Friedel-Crafts reaction. In this reaction, a coupling reaction which involves an…
Q: Can you please include the species!
A: Step 1: Step 2: Step 3: Step 4:
Q: Please explain mechanism. G.
A: Step 1: Step 2: Step 3: Step 4:
Q: OCH3 1. NaOCH3 2. HCI
A: It is the process of conversion of an ester to another ester with the exchange of groups is known…
Q: QUESTION 3 In order to induce precipitation of the salt, you need to add acid to the aqueous…
A: Answer:Salts are made during the reaction of acid and base where anion of acid combines with cation…
Q: Payli
A: The objective of this question is to find the ratio of the Fe2+ concentrations in the two half-cells…
Q: F NO2 + NaOCH2CH3
A: Given reaction:Sodium ethoxide (NaOCH2CH3) with p-fluoro nitrobenzene (1-fluoro-4-nitrobenzene)We…
Q: Suppose a 500. mL flask is filled with 0.60 mol of N2 and 0.10 mol of NO. The following reaction…
A: We will first calculate the concentration(molarity) of given species. and using the ICE table we…
Q: 2 Cr(s) + 3 Pb²⁺(aq) ⟶ 3 Pb(s) + 2 Cr³⁺(aq), what is the value of n in the Nernst equation?
A: The objective of the question is to determine the value of 'n' in the Nernst equation for the given…
Q: Can you include the species please!
A: KClO3 is a salt which is produced from the reaction between KOH (strong base) and HClO3 (strong…
Q: 3. Fill in the compounds for each step along this path in the boxes. HBr NaNH, a) NaNH2 b) CH3CH2Br…
A:
Q: List carbon-carbon bond formation reactions to synthesize 1-bromohexane from starting materials…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw the missing organic structures or select the missing reagents in the following multistep…
A: Methanol (CH3OH) acts as solvent. The E2 mechanism, where E2 stands for bimolecular elimination,…
Q: 14. Why does the rate of the reaction decrease over time? A. Exothermic reactions lose heat which…
A: Write the general rate law form. The order with respect to each reactant is not known.For the…
Q: None
A: Step 1: Basic discussion Step 2: Mechanism of the reaction Step 3:
Q: None
A: Reaction 1: In this reaction, OH group have lone pair of electrons then they attack on the carbonyl…
Step by step
Solved in 2 steps with 2 images

- Hi, I need help to perform a retrosynthetic analysis and then to design a synthesis plan. Can you answer my question in more detail? In the RSA should all disconnections, synthons, and synthetic equivalent be shown. Can you do that step by step?show the missing reagents and intermediates in the reaction sequence belowWrite down the starting materials and condition required for the retrosynthesis reaction shown below:
- Show the mechanism and reactant needed to produce these compoundsChemistry Show retro-synthetic analysis leading from the target molecule to the precursor molecule showing all intermediate molecules.Using organic chemistry from your textbook show an efficient synthesis of your target molecule starting from its parent molecule and showing the structure of each isolable intermediate.Assume you have access to usual reagents like Br2, AlCl3, Fe, HBr, HNO3, H2SO4, etc.Assume that each intermediate can be purified from other products at each step of the synthesis.For the following reactions, fill in the appropriate product(s). BE SURE TO INCLUDE ANY APPROPRIATE STERECHEMISTRY. Then, circle one criterion from each of the two columns on the right.. make sure each section includes a structure, regioselectivity criterion, and stereoselectivity criterion. You may use the image below as a template for your answer
- Perform a retrosynthetic analysis of the below including all necessary steps and reagents.draw the product of the reaction shown below. Use wedge and dash bonds to indicate stereochemistry where appropriate. Ignore inorganic byproducts.Draw the products and necessary reagents of the three step retrosynthetic reaction sequence shown below. Use wedge and dash bonds to indicate stereochemistry where appropriate. Ignore inorganic byproducts. Please choose from the following options for each of the reagents. (top reagents options) A. HNO3, cat. H2SO4 B. SO3, cat. H2SO4 C. CH3C(=O)Cl, AlCl3 D. Cl2, FeCl3 E. H2O, HCl (reagents option to the right) (top reagents options) A. HNO3, cat. H2SO4 B. SO3, cat. H2SO4 C. CH3C(=O)Cl, AlCl3 D. Cl2, FeCl3 E. H2O, HCl
- What are the starting materials you need for reaction 1 for the included molecule? Find a commercial source and report the names and CAS-numbers of the compounds. I also included the retrosynthesis for the molecule and reaction 1 so the answer is based off those.answer all of them please, thank you. Complete the following reactions in sequential order. Show the product after each step and put a box around the final productShow the reagents and intermediate products in the forward direction

