thermometer A 53.6 g sample of brass, which has a specific heat capacity of 0.375 J-g.°C', is put into a calorimeter (see sketch at right) that contains 300.0 g of water. The temperature of the water starts off at 19.0 °C. When the temperature of the water stops changing it's 20.2 °C. The pressure remains constant at 1 atm. insulated container water Calculate the initial temperature of the brass sample. Be sure your answer is rounded to 2 significant digits. sample - a calorimeter Continue
thermometer A 53.6 g sample of brass, which has a specific heat capacity of 0.375 J-g.°C', is put into a calorimeter (see sketch at right) that contains 300.0 g of water. The temperature of the water starts off at 19.0 °C. When the temperature of the water stops changing it's 20.2 °C. The pressure remains constant at 1 atm. insulated container water Calculate the initial temperature of the brass sample. Be sure your answer is rounded to 2 significant digits. sample - a calorimeter Continue
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 61E: A 5.00-g sample of aluminum pellets (specific heat capacity = 0.89 J/C g) and a 10.00-g sample of...
Related questions
Question
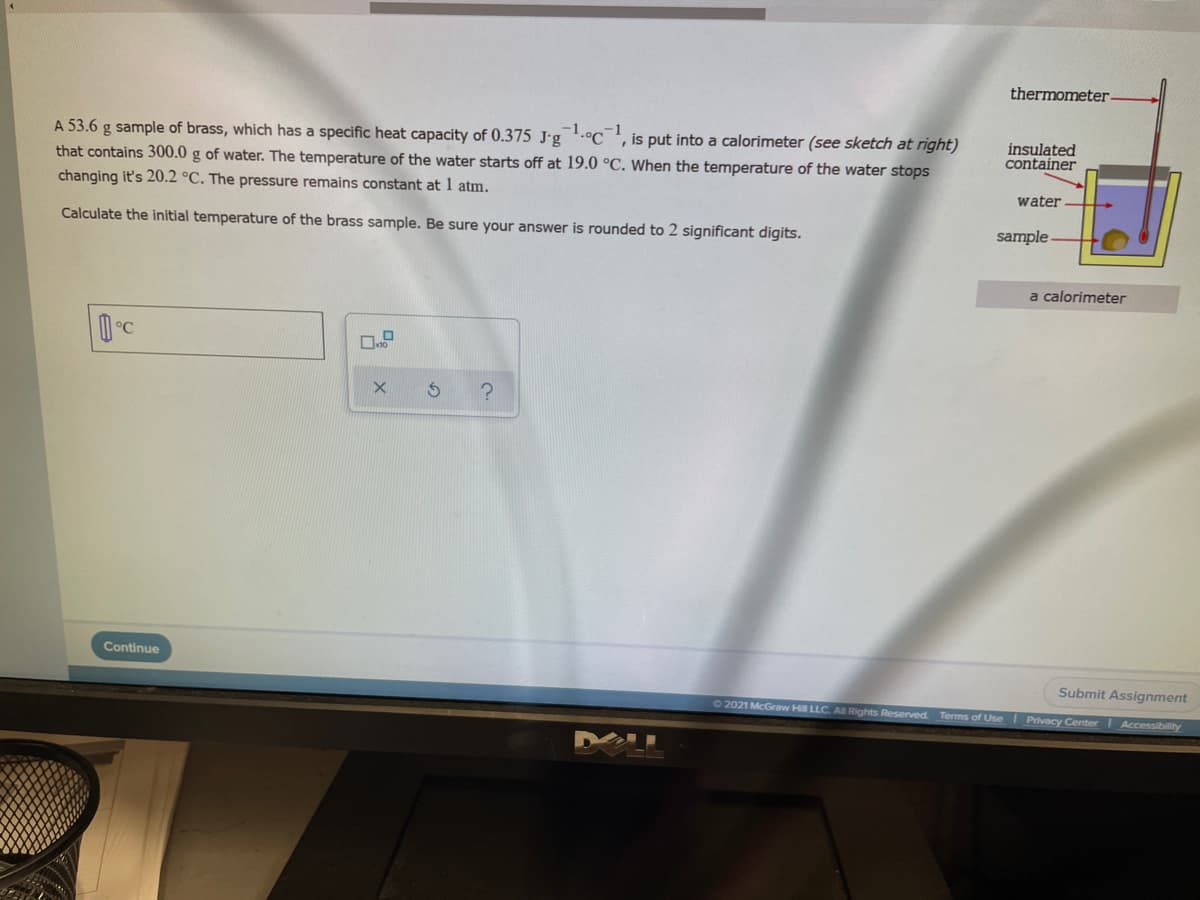
Transcribed Image Text:thermometer
A 53.6 g sample of brass, which has a specific heat capacity of 0.375 J'g.°C', is put into a calorimeter (see sketch at right)
that contains 300.0 g of water. The temperature of the water starts off at 19.0 °C. When the temperature of the water stops
changing it's 20.2 °C. The pressure remains constant at 1 atm.
insulated
container
water
Calculate the initial temperature of the brass sample. Be sure your answer is rounded to 2 significant digits.
sample -
a calorimeter
Continue
Submit Assignment
02021 McGraw Hill LLC A Rights Reserved.
Terms of Use Privacy Center I Accessibility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning