This will yield a_Straight line with slope 8. Enter the values used in the table. indicate units T(C) tave (s) k(5") T (K) 1/T (K-1) In k 20°c 293K 3.41x -l6.20 гоom 61.3s/2.05x1o 1- temperature 32°| 295 |ろ62m0 3056 |3.28x6-5.62 2025 5.2x10 283k warm water bath 3.53xió-7.54 cool 10°C water bath Use a graphing calculator or graphing program to make the graph needed to determine Eg. Sketch the graph to the right → with labeled axes and scales Ink with units. Plot the data points and draw the line of best fit. State the slope of the line. 9. Determine the experimental value of Ea with correct significant figures and units. Show your work. こmX+b Ink=In A-Ea %3D %3D -7.54 Conclusion
This will yield a_Straight line with slope 8. Enter the values used in the table. indicate units T(C) tave (s) k(5") T (K) 1/T (K-1) In k 20°c 293K 3.41x -l6.20 гоom 61.3s/2.05x1o 1- temperature 32°| 295 |ろ62m0 3056 |3.28x6-5.62 2025 5.2x10 283k warm water bath 3.53xió-7.54 cool 10°C water bath Use a graphing calculator or graphing program to make the graph needed to determine Eg. Sketch the graph to the right → with labeled axes and scales Ink with units. Plot the data points and draw the line of best fit. State the slope of the line. 9. Determine the experimental value of Ea with correct significant figures and units. Show your work. こmX+b Ink=In A-Ea %3D %3D -7.54 Conclusion
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 16QAP: Computers are not supposed to be in very warm rooms. The highest temperature tolerated for maximum...
Related questions
Question
Q. 9 is the one I need help with
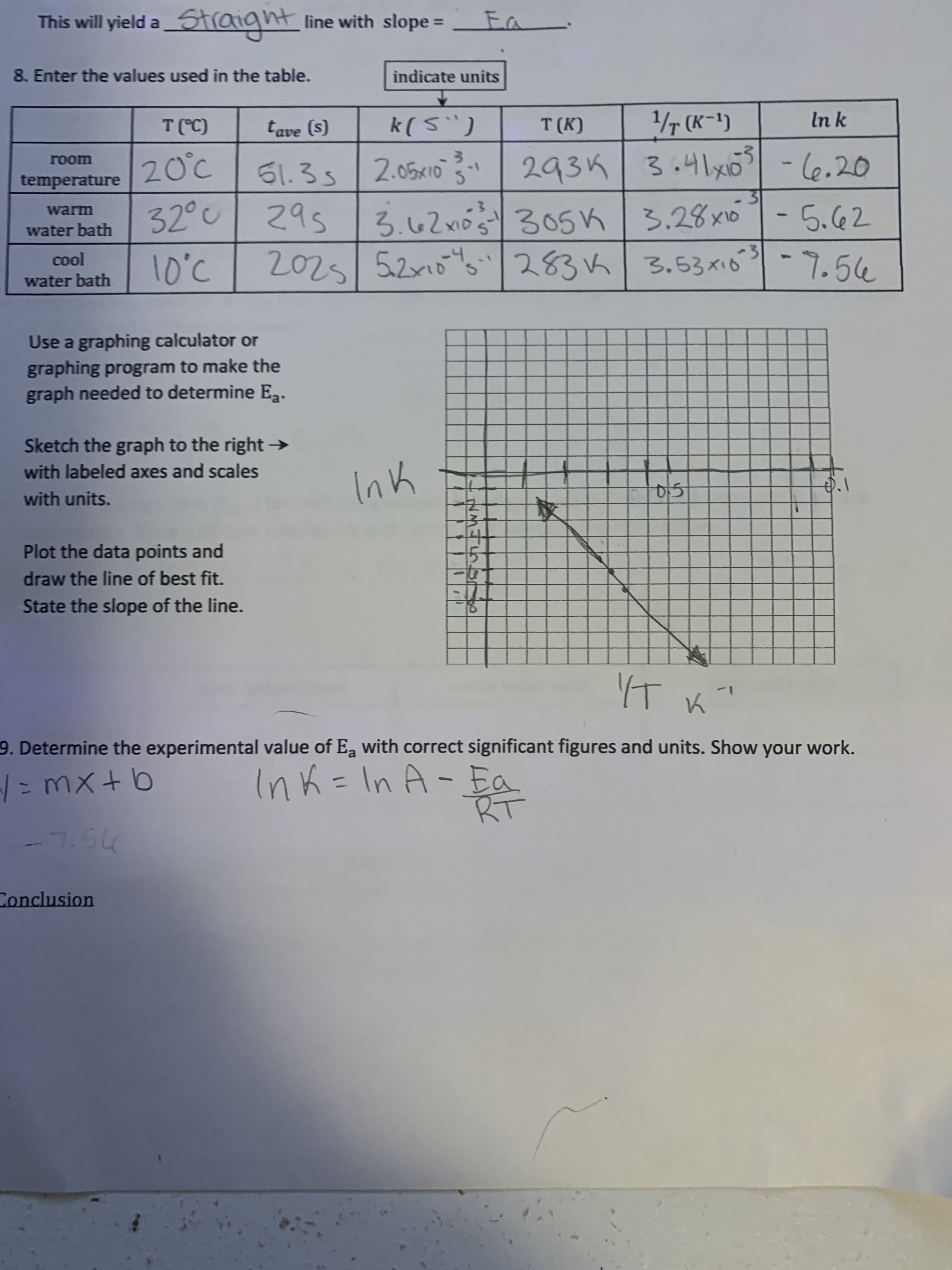
Transcribed Image Text:This will yield a_Straight line with slope
8. Enter the values used in the table.
indicate units
T(C)
tave (s)
k(5")
T (K)
1/T (K-1)
In k
20°c
293K 3.41x
-l6.20
гоom
61.3s/2.05x1o
1-
temperature
32°| 295 |ろ62m0 3056 |3.28x6-5.62
2025 5.2x10 283k
warm
water bath
3.53xió-7.54
cool
10°C
water bath
Use a graphing calculator or
graphing program to make the
graph needed to determine Eg.
Sketch the graph to the right →
with labeled axes and scales
Ink
with units.
Plot the data points and
draw the line of best fit.
State the slope of the line.
9. Determine the experimental value of Ea with correct significant figures and units. Show your work.
こmX+b
Ink=In A-Ea
%3D
%3D
-7.54
Conclusion
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning