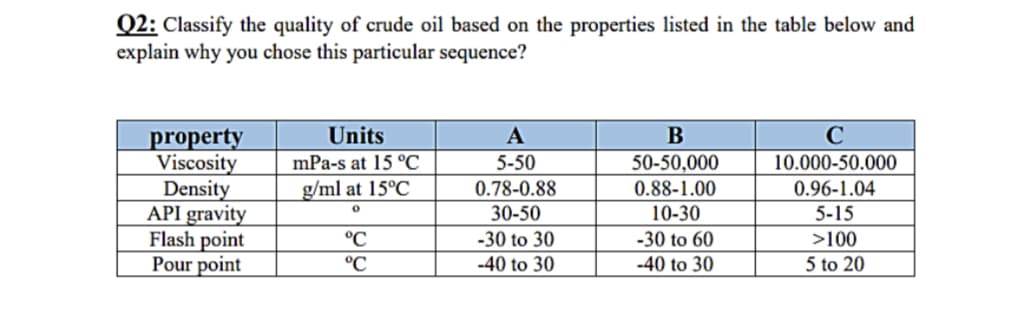
Q2: Classify the quality of crude oil based on the properties listed in the table below and explain why you chose this particular sequence? Units A B C property Viscosity mPa-s at 15 °C 5-50 50-50,000 10.000-50.000 Density g/ml at 15°C 0.88-1.00 0.96-1.04 0.78-0.88 30-50 0 API gravity 10-30 5-15 Flash point °C -30 to 30 -30 to 60 >100 Pour point °℃ -40 to 30 -40 to 30 5 to 20
Q: Students are required to keep a neat and accurate record of their laboratory observations. Upon the…
A: Group II cations: Hg2+, Bi3+, Pb2+, Cu2+, Cd2+, As3+, Sn2+, Sb3+Group reagent: H2SGroup II-A: Hg2+,…
Q: how to prepare 10% sucrose soljtion from 10gs of sucrose and 90ml of PBS?
A: Mass of sucrose = 10 g Volume of PBS = 90 ml
Q: 11) How many moles of NaNO, will be left in solution at 20 °C if a solution at 60 °C in which there…
A: Here molecular mass of NaNo3 is 84.9 g/mol Volume of solution means volume of water is 550 ml We…
Q: The following data show the rate constant of a reaction measured at several different temperatures.…
A: Frequency factor, A and activation energy barrier, Ea can be determined from the integrated form of…
Q: The Lewis Dot Structure rule states that S=N-A. Recall that S represents the TOTAL number of valence…
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a…
Q: calculated
A: Part 1: We know that molarity is defined as the number of moles of solute present in per liter of…
Q: c. Describe the steps you would follow to make 100 ml. of the 0.01 M NaHCO solution.
A: The molarity of a solution is the ratio between the number of moles of the compound to the solution…
Q: What are the M.wt of g(NH3)2.4H2O if know the atomic .mass are H= 1, O= 16
A:
Q: Nitrous acid is a weak acid and has a concentration of 0.65 mol/L. What is the pH of the solution…
A:
Q: The molar solubility of PbBr2 is 2.17 x 10-3 M at a certain temperature. Calculate Ksp for PbBr2.
A:
Q: 4. Draw the two chair conformation for Galactose. Tell which one is more stable, and why? Assign R…
A: Chair confirmation : A six-membered ring confirmation in which atoms 2,3,5 and 6 lie in the same…
Q: OH H₂SO4 HỌC–CH2–CH2CH3 HỌC=CH–CH2CH3 H₂O Acid-catalyzed dehydration of 1-butanol yields 1-butene as…
A: The acid catalysed dehydration of alcohol has 3 steps- 1- protonation of OH group 2- removal of…
Q: (9.7)Which of the following is diamagnetic? O Sn²+ O Cr ΟΝ OF
A: Given, Which of the following is diamagnetic ? Options are : a. Sn2+ b. Cr c. N d. F
Q: Balance the following chemical equation by providing the correct coefficients. [?]Pb(NO3)2 + Na₂CrO4…
A: In the balanced chemical equation, the number of all the atoms or elements are equal in both…
Q: Which is a primary amine! O (CH₂CH₂)2N © CHINH O (CH3)3CNH₂ O(CH₂4N*CT
A:
Q: 5.) What is the difference between -OH and OH-?
A:
Q: What is the percent by volume concentration of a solution in which 75.0 ml of ethanol is diluted to…
A:
Q: Which of the following compounds will undergo radical bromination (using Br₂ and light) with the…
A:
Q: 500 mL of a 1.52 mol/L solution of vinegar (acetic acid) is used to react with a 2.53 g sample of…
A: Given the molarity of acetic acid(CH3COOH) in the vinegar solution, C = 1.52 mol/L Volume of acetic…
Q: 2. Write the Lewis dot symbols of the reactants and products in the following reactions. (First…
A:
Q: Rimantadine was among the first antiviral drugs to be licensed in the United States to use against…
A: Experimental condition to achieve step 3 product would be by reacting with sodium amide in the…
Q: "art A What mass of phosphorous pentafluoride has the same number of fluorine atoms as 27 grams of…
A:
Q: 5. Which of the following statements is correct? A. The rate of a reaction is independent of…
A: 26) here answer d is correct that rate of reaction decreases with time and concentration of reactant…
Q: Refer to reaction sequence below then answer the following question: oxidation CH,OH oxidtion The…
A: The oxidation of alcohol depends on the number of hydrogen atoms attached to the carbon atom that…
Q: a) H c) d) с C 1 H Н H b) Н H Н н Н I I I I 1 H-C с c-c-c-H I I I Н Н ОН Н H CI CI н-с-с-Н 1 I CI CI…
A:
Q: The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an…
A:
Q: Part A What mass of phosphorous pentafluoride has the same number of fluorine atoms as 27 grams of…
A:
Q: Phosphoric acid, H3PO4(aq), can be used to remove rust. Find the molar concentration of an 85% (m/v)…
A:
Q: The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer…
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Draw the major product of this reaction. Ignore any inorganic byproducts. HBr (1 oqui
A:
Q: A mass of TAG (2.151 g) was weighed out accurately. It was then subject to reflux with 100.0 mL of…
A: Saponification value :- The milligram of KOH required to neutralize fatty acid resulting (formed)…
Q: The electron in a Bohr hydrogen atom is bound less tightly when it has a quantum number of 3 than…
A:
Q: 8. Below you will find a molecule with protons labeled a-j, match the corresponding peak to its…
A:
Q: 7. Explain how the molar conductivities can be used to generate plots to determine the acid…
A: To get the acid dissociation constant, plot a graph between reciprocal of concentration of acetic…
Q: 15 kg a perfect gas is heated at constant pressure from 15 °C to 368.15 K, the heat required is 1130…
A:
Q: 17.A solid piece of limestone (calcium carbonate) is reacted with 1.0 mol/L sulfuric acid at 25 °C.…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 10 A chemist conducts an experiment in which 2.00 L of hydrogen gas is collected over water at 1.00…
A: #10: #(a): Temperature, T = 298.15 K The saturated gas contains both dry H2(g) and water vapor,…
Q: If 0.530 moles of gas are removed from the initial 8.11 moles and the temperature increased to 52.8…
A:
Q: Calculate the mean activity coefficient y, for the Ba2+ and SO2 ions in a saturated solution of…
A:
Q: a)What is the conjugate base of HXO4 −(aq)? a H3O +(aq) b OH −(aq) c H2XO4(aq) d HXO4 −(aq) e…
A: Here, we have to find the conjugate base of HXO4-. Also, we have to find the concentration of a weak…
Q: 33. Which of the following statements is true for the shown reaction? --CH P-O-CH₂ CH₂OH CH₂OH HO OH…
A: Even in non-photosynthetic animals, gluconeogenesis is a metabolic route that leads to the…
Q: At 277.0 K, a gas has a pressure of 99.5 kPa. What is the pressure of the gas at 210.0 K, if the…
A:
Q: Find the percent by mass in which 41.0 g of NaCl (58.445 g/mol) is dissolved in 331 grams of water
A:
Q: Calculate the concentration of HC6H₂O6 in an aqueous solution of 0.0337 M ascorbic acid, H₂C₂H₂O6…
A: Concentration of ascorbic acid (H2C6H6O6) = 0.0337 M Concentration of HC6H6O6- = ?
Q: Account for the regioselectivity and stereoselectivity observed when this compound is treated with…
A: Oxymercuration reaction of alkenes follows Markovnikov's rule in which the nucleophile attacks at…
Q: Procedure 1: Construct a conductivity meter and test Solutions Conductivity (None, Dim, or Bright)…
A: Generally metals , solid ionic compound's solution shows conductivity and solid compound and…
Q: Ithy Alternative X BU Homepage INTRC xb Success Confirmati x Assignments a…
A: Endothermic reactions are those reactions in which there is absorption of heat energy.
Q: 1. Which of the following is the correct name for the structure below? a. trans-4-nonyne c.…
A:
Q: What is the oxidation state on the central carbon in the following compound? H₂C-CH
A:
Q: Q1. The colour of silver and gold nanoparticles changes according to the particle's ....... OK
A: The color changes depending on the size/shape of the gold nanoparticles.
Step by step
Solved in 3 steps with 2 images

- A core sample is saturated with an oil (?o = 35oAPI), gas and water. The initial weight of the sample is 224.14 g. After the gas is displaced by water (?w = 1 g/cm3 ), the weight is increased to 225.90 g. The sample is the placed in a Soxhlet distillation apparatus, and 4.4 cm3 water is extracted. After drying the core sample, the weight is now 209.75 g. The sample bulk volume, 95 cm3 is measured in a mercury pycnometer. Find the porosity, water saturation, oil saturation, gas saturation and lithology of the core sample.There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…
- In a solution containing 25ml (NH4)2S2O8 with a total volume = 100 ml =0.1 liters , the solution composition are shown below.Note; the solution require 25ml of 0.2M S2O8^-2.SOLUTION Kl KNO3 EDTA Na2S2O3 STARCH50ml 23ml 1 drop 1 ml 10 dropsExperiment; initial (S2O8-2) =0.05M ; INITIAL (l-)=0.10 Mml of S2O3^-2 added Time in minutes and seconds for color change Cumulative time in seconds Total moles of S2O8^-2 consumed1 1:43 2.0 x 10^-42 1:20 4.0 x 10^-43 1:13 6.0 x 10 ^-44 1.17 8.0 x 10 ^-45 1.24 10 x 10 ^-41a) Find the cumulative time in secondsb) for the runs ,plot mole of S2O8^2- REACTION vs. time in seconds2) Draw a straight line through ( origin ) the points and calculate the slope3) divide the value of the slope by the total volume of ( 0.1 L) to get the rate in units of M/s4) find the rate5 )calculate x and y and kGiven that liquid alum is used as a coagulant. Specific gravity of alum is 1.33. One gallon ofalum weighs 11.09 pounds (5.03 kg) and contains 5.34 pounds (5.42 kg) of dry alum.Determine: (a) mL of liquid alum required to prepare a 100 mL solution of 20,000 mg/L alumconcentration, (b) the alum concentration, (c) the dosage concentration of 1 mL of stocksolution in a 2000 mL Gator jar sample.• Determine alum concentration in mg/L• Prepare 100 ml stock solution having a 20,000 mg/L alum concentration• Calculate mL (y) of liquid alum to give 2000 mg• Find 1 mL of alum concentration (z) in 2000 mL sample (jar)Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/LCharge balance equation:[H+] = [OH-] + [HCO3-] + 2[CO32 ] If the CO2 concentration in the atmosphere is 300 ppm, what is the pH of the ocean?
- What is the molarity of a concentrated solution of sulfuric acid with a specific gravity of 1.84 and 95.0% by weight sulfuric acid? specific gravity = density of substance at certain temp/density of water at 4 degrees C the density of water is 0.999973 g/mL at 4 degrees celsius. Describe how to prepare 3.0 L of 0.100M. I will pipet ______ and add this to 200mL water. The solution will be stirred and diluted to 3L with water.You take a representative soil sample and determine the following data: Exchangeable cation cmolc/kg of soil Ca2+ 2.7 Mg2+ 1.0 K+ 0.1 H+ 1.0 Al3+ 2.8 Na+ 0.1 1) What is the CEC (cmolc/kg ) of this soil?The alcohol content of hard liquor is normally given in terms of the “proof,” which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the number of grams of alcohol present in 1.00 L of 75-proof gin. The density of ethanol is 0.798 g/mL. Round off to 4 sig. figures for all calculations made prior to final answer and the final answer should be in 3 sig. figures
- Prepare 1500mL of 0.5N H2SO4 solution with specific gravity of 1.84g/mL and assay of 97%I asked this question earlier but I think I forgot to include a conversion wht we know : mm of citrix acid = 192.14g/moldensity of citrix acid =166g/cm3 1 lime = 2tbsp of juice = 6 tsp 1tsp = 4.929x10-3L Q: assume citrix acid makes up 7.96% by volume of the lime juice . How many moles of citrix acid are in the recipe ( calls for 1 lime juice)1. You give your home health patient an unopened 500-mL bottle of guaifenesin and tellthem to take 2 teaspoons 4 times a day as ordered. They ask you how long the bottlewill last. 2. You are shadowing a nurse during a clinical who receives an order to adjust the infusionrate of a pump so that 1.6 mg of lidocaine are being delivered per minute. Hanging is a100 mL piggyback containing 0.4 grams lidocaine, a 0.4% solution. How many mL perhour should the piggyback be set to deliver? How long will the piggyback last? 3. Phenobarbital 180 mg/m 2 /24 hours given every eight hours is ordered for a childwhose BSA (body surface area) is 0.29 m 2 . How many mg will you need if the child willbe on the medication for 10 days? 4. You are going to mix a 1:10 bleach solution with water to make 500 mL of a 1:35 bleachsolution. How much 1:10 bleach solution should you use?(I recommend using the C1V1=C2V2 formula) 5. As a home health nurse you need to help a client make…

