Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.15QAP
Related questions
Question
6
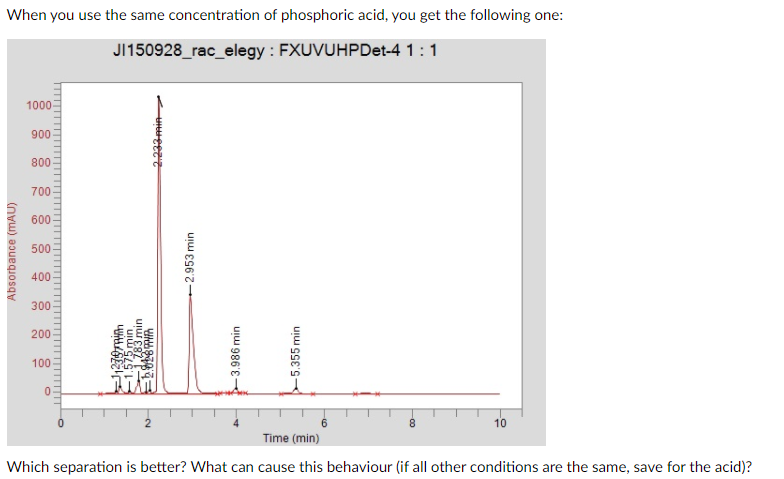
Transcribed Image Text:When you use the same concentration of phosphoric acid, you get the following one:
JI150928_rac_elegy : FXUVUHPDet-4 1:1
1000
900
800-
700
600
500
400
300
200
100
8.
10
Time (min)
Which separation is better? What can cause this behaviour (if all other conditions are the same, save for the acid)?
Absorbance (mAU)
Uju E82
-2.953 min
3.986 min
5.355 min
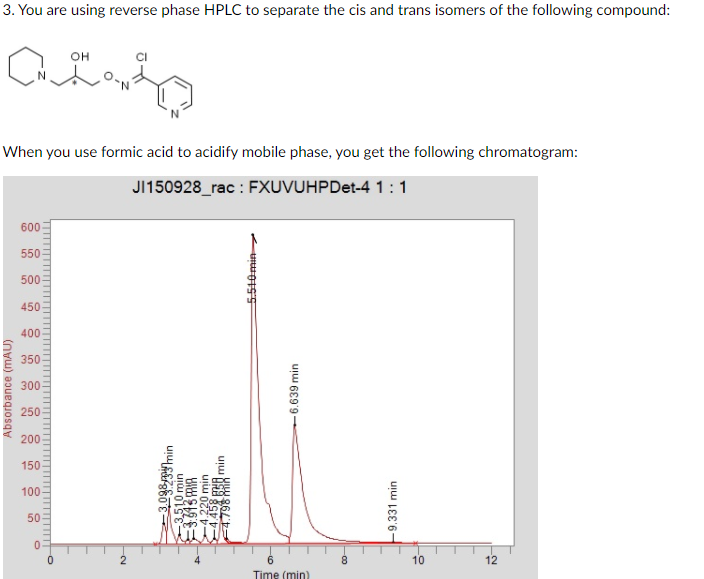
Transcribed Image Text:3. You are using reverse phase HPLC to separate the cis and trans isomers of the following compound:
он
When you use formic acid to acidify mobile phase, you get the following chromatogram:
J150928_rac : FXUVUHPDet-4 1:1
600
550
500
450
400
350
300
250
200
150
100
50
10
12
Time (min)
Absorbance (mAU)
3,098 gimin
3,510 min
913 MIA
-4.220 min
BE min
5.540min
-6.639 min
9.331 min
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT