Titration curve of tripeptide HAY 14 13 12 11 10 7. 10 15 20 25 30 Volume 0.1M NaOH titrated Hd
Titration curve of tripeptide HAY 14 13 12 11 10 7. 10 15 20 25 30 Volume 0.1M NaOH titrated Hd
Chapter1: Numerals And Fractions
Section: Chapter Questions
Problem 6PP
Related questions
Question
Indicate the Amino-pKa and the pI value of the HAY tripeptide on the titration curve.
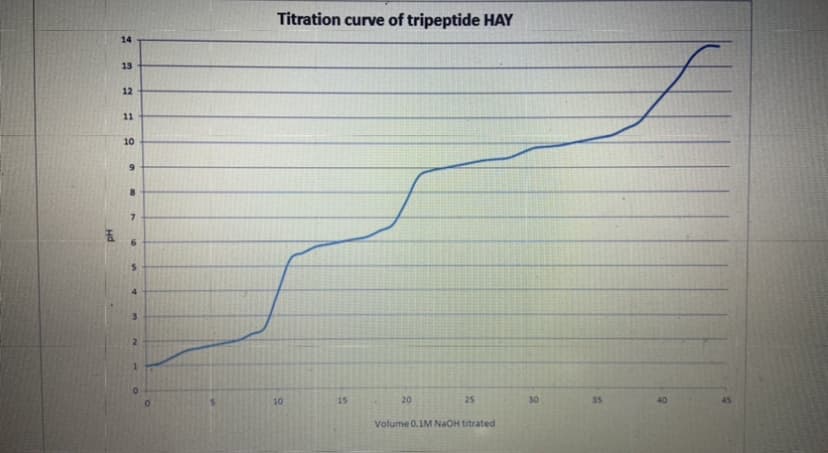
Transcribed Image Text:Titration curve of tripeptide HAY
14
13
12
11
10
10
15
20
25
30
35
40
45
Volume 0.1M NaOH titrated
Hd
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning