Q: A student ran the following reaction in the laboratory at 617 K: Co(g) + Cl2(g) = cocl2(g) When she ...
A:
Q: A chemist dissolves 203. mg of pure barium hydroxide in enough water to make up 60. mL of solution. ...
A: We have to predict the pH of the solution.
Q: 7.53 Calculate th 20°C if the pressi radius is 0.040 cm
A:
Q: NH,NO2 NH,I NaOH NaClO Nal
A:
Q: 1. For substances that will be in thermal contact, how would we know which will absorb energy (heat)...
A: #1: When two substances are in thermal contact with each other, heat is transferred in the form of t...
Q: Draw the Lewis Dot Symbol of the following atoms or ions. 1. Radium 2. Selenium 3. Gallium
A:
Q: ature (Tg) is the temperature range where a polymer chain gains segm y of the polymer chains depends...
A: The glass transition temperature (Tg) is the temperature at which a polymer turns from a ductile mat...
Q: Hypochlorous acid, HOCI(aq), is a weak acid (Ka = 4.0 x 10-8) used in the chlorination of drinking w...
A: a.) To calculate pH , we would draw an ICE table and use its Ka value. b.) Percent ionization is Ca...
Q: Which highlighted bond requires the highest energy to break? Lowest? In between? Answer these questi...
A:
Q: CH2 CH; e. CH,=CHCCH; с. a. CH;CHCH=CH2 ČH3 ČH3 CH3 CH3 d. f. b. CH3CHCH2CH=CH2 CH3 What is the majo...
A: When alkene compound is treated with HBr then Markovnikov's addition reaction takes place via carboc...
Q: Which is the correct Ksp expression for Cr(OH)3(s) dissolving in water?
A: we need to write the expression for Ksp for Cr(OH)3(s)
Q: If 0.25 moles of HCL were mixed with 0.25 moles of NaF (in 1.0 L final volume), what would be the pH...
A: pH can be calculated from the concentration of total H+ ions present in the solution. pH = - log [H+...
Q: el). Based on your answer, draw two lines on one reaction coordinate diagram that show the relative ...
A: We have to answer the following questions.
Q: How to interept this data to match the structure? It gives the peak/PPM and having trouble matching ...
A: The given NMR data is needed to be interpreted and the each signal obtained is needed to be assigned...
Q: 0 30 5060 70 3600 UALA Weight percent tungsten 90 80 100 3422°C 3400 - 3200 3000 2800 2665 C 2670 C ...
A: Few questions based on phase diagram that are to be accomplished.
Q: predicting the IR spectrum for the product with specific absorbtion bands and for specific funstiona...
A:
Q: Many ions do not have tabulated activity coefficients nor can the activity coefficient be estimated,...
A:
Q: Please answer all questions below with calculations. 1. What is the empirical formula of a hydrocar...
A: Since you have asked multiple questions questions, we will solve the first question for you. If you ...
Q: What quantity in moles of precipitate will be formed when 100.0 mL of 2.00 M NaCl is reacted with 72...
A:
Q: iron (III) oxide carbon monoxide → iron carbon dioxide
A: Iron(III)oxide + carbon monoxide ---> Iron + carbon dioxide Formula and chemical reaction is ---...
Q: Determine the concentration (M) of H3O+ in a 0.577 M solution of a triprotic weak acid, H3A, given t...
A: Here we have to determine the pH of 0.577M triprotic acid H3A . Given Ionization constants are Ka1= ...
Q: Iron metal reacts with oxygen to produce iron (III) oxide. Write the Chemical Equation First. A.) Ho...
A:
Q: When a small amount of strong acid is added to an ammonia/ammonium chl buffer, what happens to the c...
A: Given : We have to tell what happens to the concentration of ammonia and ammonium chloride buffer wh...
Q: onsider the following system at equilibrium where AH° = -87.9 k), and Cl3(g) + Cl2(g) =PCI5(g) the V...
A:
Q: A gas occupies 6.5 L at 0 °C and 760. torr. What volume does this gas occupy at STP? a. 3.3 L b...
A: Given:: P1=760 torr, V1=6.5 L , T1 = 0℃ = (0+273 K)= 273 K V2= ?, P2= 1 atm = 760 torr (STP) T2 = 27...
Q: What can affect the formation of different crystalline phases
A: A question based on phase diagram that is to be accomplished.
Q: In acidic solution: 1. PbO2 + I2 Pb2+ + IO3 Oxidation half-reaction: Reduction half-reaction: Comple...
A:
Q: Consider the following reaction where K, = 10.5 at 350 K. 2CH2CI2(g) CH4(9) + CCI4(g) A reaction mix...
A:
Q: What is the principal quantum number (n), the orbital angular momentum quantum number (l), the magne...
A: Given- Boron (B) Atomic number = 5
Q: Trial 1 Trial 2 Mass of ice Mce (grams) 79 79 Mass of warm water 120 119 Mwater (grams) Initial temp...
A: Heat lost by substance at higher temperature is equal in amount to the heat gained by the cold body ...
Q: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric acid. Usin...
A: Given: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric aci...
Q: What is the pOH of the solution in Part B? Express the poH numerically to two decimal places. ? pOH ...
A:
Q: you will titrate 2.00 mL of 0.0100 M KMnO4 in base with 0.0200 M NaHSO3. Using the correct redox equ...
A:
Q: What is the principal quantum number (n), the orbital angular momentum quantum number (l), the magne...
A: There are 4 quantum numbers : 1) principal quantum number (n) 2) orbital angular momentum quantum n...
Q: The equilibrium constant, Ko, for the following reaction is 1.29x10-2 at 600K. cocl,(g) co(g) + Cl2(...
A: Given, Number of moles of COCl2(g) = 0.265 mol Volume = 1 L Molarity = number of moles/volume in...
Q: a. NH2 + H2N CI hexamethylene diamine b. CI CH2 vinyl chloride
A: The reactions given are,
Q: Protectim of C=0 TSOH benzene HO reglux TS OH =
A: In presence of acid diol can protect carbonyl group.
Q: Using activities, calculate the pH of a solution containing 0.015 M HCI, 0.036 M HCI and 0.050 M NaN...
A:
Q: Write balanced half-reactions for the following redox reaction: Cr,0; (aq)+7H,0(1)+3 Zn(s) → 2Cr**(a...
A: To solve this problem we have identifie which species is oxidised and which is reduced .
Q: 10. Which of the following is a cis-isomer or a trans-isomer? H. H. Br a. CH H. C. White the initiat...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: Q2: Calculate the solubility of Mg(OH)2 in 0.10 M NaOH.Calculate the solubility in grams/liter (use ...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Draw the correct lewis structure for the following molecule. NC
A:
Q: Ethanol can be created by reacting hydrogen gas, carbon, and oxygen together. Using the following th...
A: The reactions given are,
Q: 8+83= %3D Oxygen Carbon %3D
A: Combination reaction: A combination reaction is a type of chemical reaction in which two reactants c...
Q: Three students are preparing alkoxides in the lab by mixing sodium metal with a suitable alcohol. St...
A: Rate of a reaction depends upon various factors. Here three student perform a reaction to prepare so...
Q: In acidic solution: 1. PbO2 + I2 → Pb2+ + IO3 Oxidation half-reaction: Reduction half-reaction: Comp...
A:
Q: Lab. 7 1. ___medium and _____reagent are used to detect indole production. _____will react wit...
A: Ans. 1. medium : The medium used with this reagent is either motility- indole-ornithine agar, brot...
Q: Dinitrogen tetraoxide, a colorless gas, exists in equilibrium with nitrogen dioxide, a reddish brown...
A:
Q: Why can a molecule such as CO easily be dissociated on a metal (Au/CeO2) surface but not in the gas ...
A: Adsorption (chemisorption) on a metal surface
Q: Determine the K, of a weak base if a 0.28 M solution of the base has a pH of 10.14 at 25°C. K x 10
A: Given: Concentration of weak base (Assuming B) = 0.28 M And the pH of the solution = 10.14
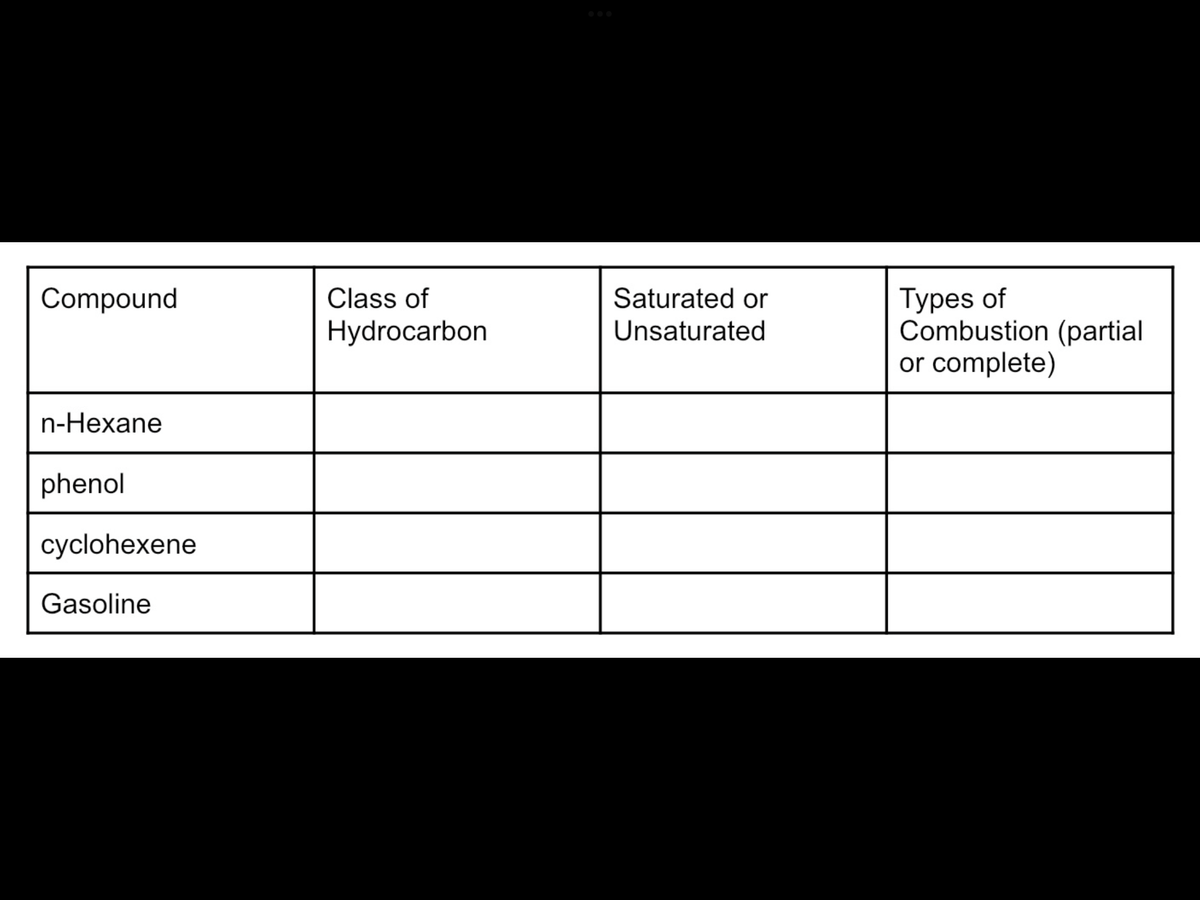
Complete the table.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Arrange the following groups in order of increasing priority. Q.) -CH3 -CH2OH -CH2NH2 -CH2BrIsomerization of (R)-Carvone What is the mechanism of the attached reaction and the final product(s)?Consider the molecule: rate the priority functional groups from highest to lowest A. Alkyl chain B. Ketone C. Carbonyl D. Anhydride
- Name the major product of the reaction between (S)-2-bromohexane and KCH3COO. Be specific with the configuration.How many isomeric dimethylcyclobutanes are thereOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- DHA is a fatty acid derived from sh oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H] and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (ve equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z conguration?trans-stilbene+pyrindium tribromide _____>(acetic acid) 1,2-dibromo-1,2-diphenylethane What are steroisomers of the product? What products will form if reaction is not stereospecific? What if reaction is sterospecifc, what products will form?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?

