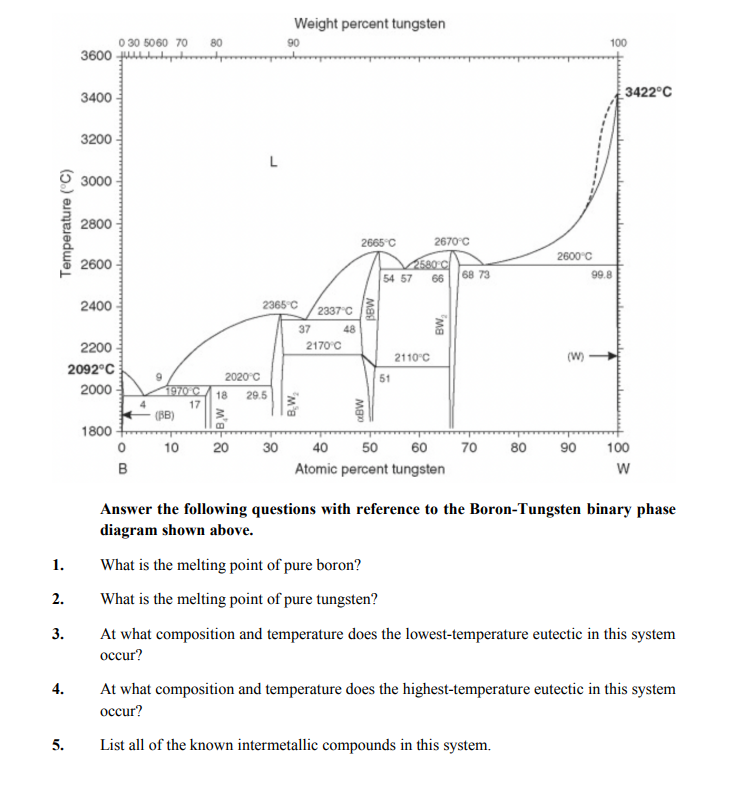
0 30 5060 70 3600 UALA Weight percent tungsten 90 80 100 3422°C 3400 - 3200 3000 2800 2665 C 2670 C 2600 C 2580 C 54 57 2600 68 73 99.8 66 2400 - 2365°C 2337 C 37 48 2200 - 2170°C 2110'C (W) 2092°C 2020°C 51 2000 1970 C 18 29.5 17 (BB) 1800 10 20 30 40 50 60 70 80 90 100 в Atomic percent tungsten Answer the following questions with reference to the Boron-Tungsten binary phase diagram shown above. 1. What is the melting point of pure boron? 2. What is the melting point of pure tungsten? 3. At what composition and temperature does the lowest-temperature eutectic in this system осcur? 4. At what composition and temperature does the highest-temperature eutectic in this system occur? 5. List all of the known intermetallic compounds in this system. Temperature (°C) B,W, BBW BW,
0 30 5060 70 3600 UALA Weight percent tungsten 90 80 100 3422°C 3400 - 3200 3000 2800 2665 C 2670 C 2600 C 2580 C 54 57 2600 68 73 99.8 66 2400 - 2365°C 2337 C 37 48 2200 - 2170°C 2110'C (W) 2092°C 2020°C 51 2000 1970 C 18 29.5 17 (BB) 1800 10 20 30 40 50 60 70 80 90 100 в Atomic percent tungsten Answer the following questions with reference to the Boron-Tungsten binary phase diagram shown above. 1. What is the melting point of pure boron? 2. What is the melting point of pure tungsten? 3. At what composition and temperature does the lowest-temperature eutectic in this system осcur? 4. At what composition and temperature does the highest-temperature eutectic in this system occur? 5. List all of the known intermetallic compounds in this system. Temperature (°C) B,W, BBW BW,
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 102GQ: Cloth can be waterproofed by coating it with a silicone layer. This is done by exposing the cloth to...
Related questions
Question
Transcribed Image Text:Weight percent tungsten
0 30 5060 70 80
3600 uLtt
90
100
3400 -
3422°C
3200
L
3000
2800
2665°C
2670°C
2600 C
2600
2580 C
99.8
54 57 66 68 73
2400
2365 C
2337 C
37
48
2200 -
2170'C
2110°C
(W)
2092°C
2020°C
51
2000
1970 C
17
(BB)
18
29.5
1800
10
20
30
40
50
60
70
80
90
100
Atomic percent tungsten
Answer the following questions with reference to the Boron-Tungsten binary phase
diagram shown above.
1.
What is the melting point of pure boron?
2.
What is the melting point of pure tungsten?
3.
At what composition and temperature does the lowest-temperature eutectic in this system
occur?
4.
At what composition and temperature does the highest-temperature eutectic in this system
occur?
5.
List all of the known intermetallic compounds in this system.
Temperature (°C)
B,W.
Mad
BW,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning
