Two amino acids are shown below. Name them in the blanks, and then draw the structure of the dipeptide they would form if a peptide bond connected them (in the order shown.) Draw the dipeptide as a Zwitterion, and name it in the blank. 118 H₂N-CH- CH3 + H₂N-CH₂-CⓇ
Two amino acids are shown below. Name them in the blanks, and then draw the structure of the dipeptide they would form if a peptide bond connected them (in the order shown.) Draw the dipeptide as a Zwitterion, and name it in the blank. 118 H₂N-CH- CH3 + H₂N-CH₂-CⓇ
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter3: Biological Molecules: The Carbon Compounds Of Life
Section: Chapter Questions
Problem 2TYK: Which of the following characteristics is not common to carbohydrates, lipids, and proteins? a. They...
Related questions
Question
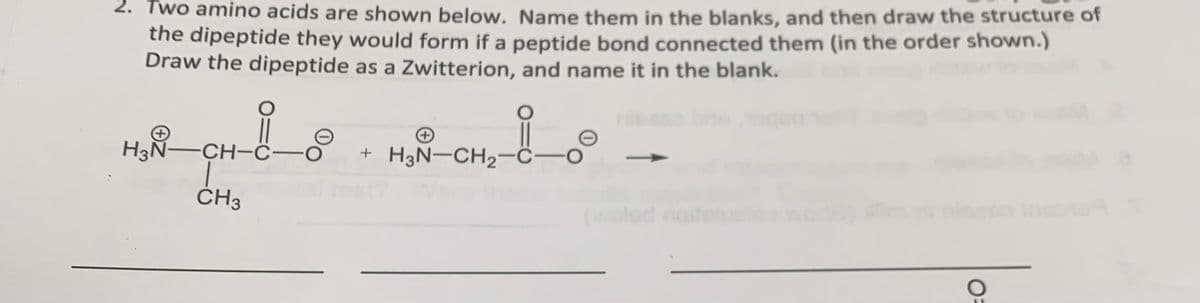
Transcribed Image Text:2. Two amino acids are shown below. Name them in the blanks, and then draw the structure of
the dipeptide they would form if a peptide bond connected them (in the order shown.)
Draw the dipeptide as a Zwitterion, and name it in the blank.
H3N-CH-
CH-C-O
CH3
+
+ H3N-CH₂-C O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning