Two different compounds are shown below. Both are soluble in water. The option that shows the predominant interactions that would explain the solubility of each of them respectively are: a) Hydrogen bonds and induced dipoles (London scattering) b) Ion-molecule forces in both cases c) Hydrogen bonds and forces ion — molecule d) Ion forces - induced dipole and induced dipoles (London scattering)
Two different compounds are shown below. Both are soluble in water. The option that shows the predominant interactions that would explain the solubility of each of them respectively are: a) Hydrogen bonds and induced dipoles (London scattering) b) Ion-molecule forces in both cases c) Hydrogen bonds and forces ion — molecule d) Ion forces - induced dipole and induced dipoles (London scattering)
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.1: Naming Cycloalkanes
Problem 2P: Draw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcyclooctane (b)...
Related questions
Question
Two different compounds are shown below. Both are soluble in water.
The option that shows the predominant interactions that would explain the solubility of each of them respectively are:
a) Hydrogen bonds and induced dipoles (London scattering)
b) Ion-molecule forces in both cases
c) Hydrogen bonds and forces ion — molecule
d) Ion forces - induced dipole and induced dipoles (London scattering)
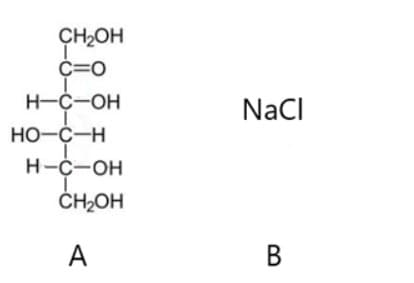
Transcribed Image Text:CH2OH
Č=0
H-C-OH
NaCI
Но-с—н
H-C-OH
ČH2OH
A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

