Type your answers in all of the blanks and submit While we haven't really discussed this yet, there is actually a mathematical relationship that links wavelength and energy. This is a very famous equation and takes the form E = hc/A where h = planks constant = 6.63 x 10-34 and c= the speed of light = 3x 108 m/s. Use this equation to determine the Energy of the following photons: 1. A photon witha wavelength of 500 nm. E = Type your answer here ! Please type your answer to submit Fullscreen 1. A photon with a wavelength of 840 nm. E= Type your answer here 11:19 PM 23 S/3/2021 Cip
Type your answers in all of the blanks and submit While we haven't really discussed this yet, there is actually a mathematical relationship that links wavelength and energy. This is a very famous equation and takes the form E = hc/A where h = planks constant = 6.63 x 10-34 and c= the speed of light = 3x 108 m/s. Use this equation to determine the Energy of the following photons: 1. A photon witha wavelength of 500 nm. E = Type your answer here ! Please type your answer to submit Fullscreen 1. A photon with a wavelength of 840 nm. E= Type your answer here 11:19 PM 23 S/3/2021 Cip
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter5: Electron Configurations And The Periodic Table
Section: Chapter Questions
Problem 31QRT
Related questions
Question
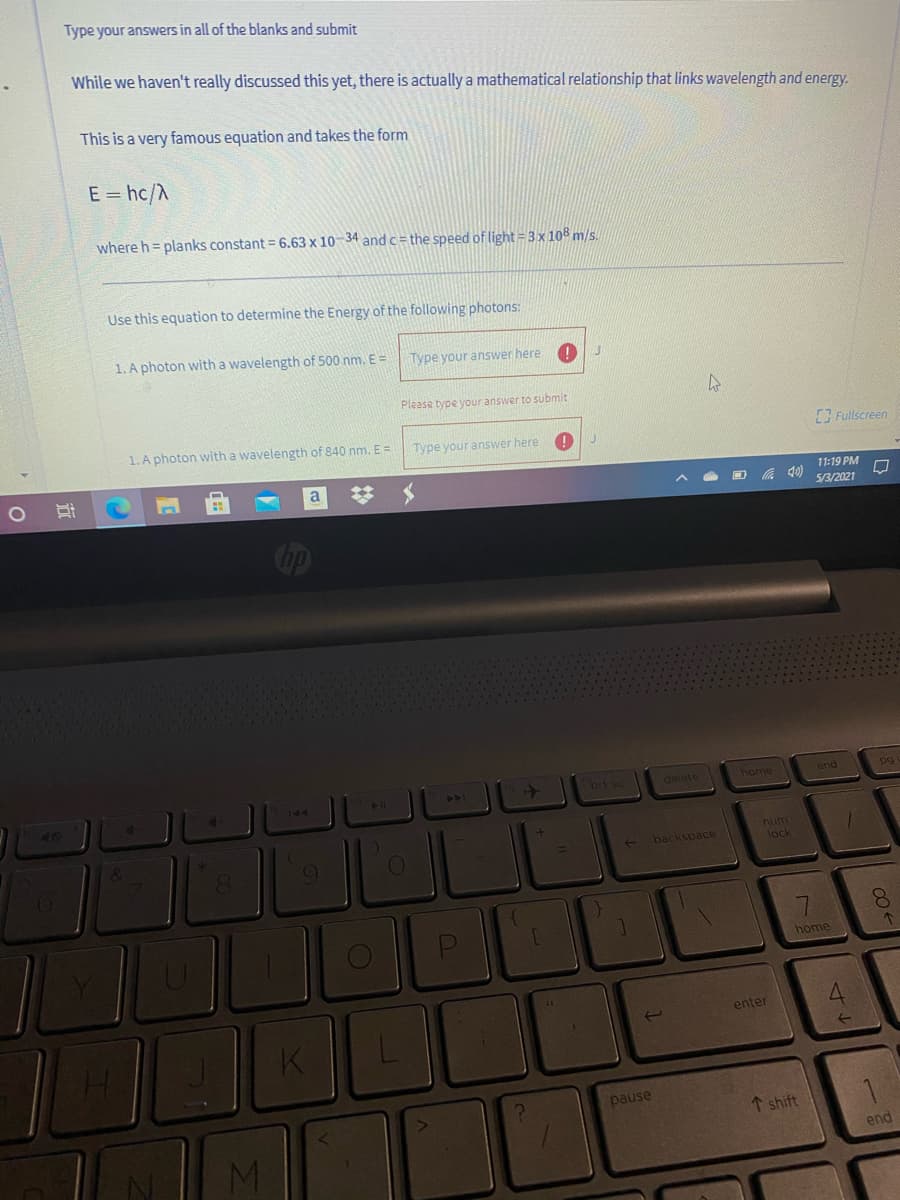
Transcribed Image Text:Type your answers in all of the blanks and submit
While we haven't really discussed this yet, there is actually a mathematical relationship that links wavelength and energy.
This is a very famous equation and takes the form
E = hc/A
where h = planks constant = 6.63 x 10-34 andc= the speed of light = 3x108 m/s.
Use this equation to determine the Energy of the following photons:
1. A photon with a wavelength of 500 nm. E =
Type your answer here
Please type your answer to submit
1. A photon with a wavelength of 840 nm. E =
Type your answer here
Fullscreen
a
%23
11:19 PM
5/3/2021
brt sc
home
delete
end
>>
144
40
num
lock
backspace
8.
home
enter
K.
1.
pause
↑ shift
end
MI
44
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning