Unit Cell and Crystal Characteristic The volume of sphere is 4/3 (7) r³ and the radius of each atom is 1.0 cm. Assume that all the solids are aluminum (MM=26.98 g/mol). Calculate the following: number of atoms(spheres) inside the unit cell, total volume of spheres inside the unit cell, volume of the unit cell and the % of space occupied in the unit cell using the equation: Density (g/cc) Vspheres Vcel % occupied Cubic (express to no. of atoms cm3 cm3 scientific Unit (whole no.) notation; round- (round-off to two | (round-off to two decimal places) Cell (round-off to two decimal places) decimal places) off to two decimal places) SC x10-24 FCC x10-24 ВСС x10-24 Based on the calculated density, what is the most probable lattice type of aluminum metal? Body Centered Cubic (BCC) Structure, Simple Cubic (SB) Structure, or Face-Centered Cubic (FCC) Structure?
Unit Cell and Crystal Characteristic The volume of sphere is 4/3 (7) r³ and the radius of each atom is 1.0 cm. Assume that all the solids are aluminum (MM=26.98 g/mol). Calculate the following: number of atoms(spheres) inside the unit cell, total volume of spheres inside the unit cell, volume of the unit cell and the % of space occupied in the unit cell using the equation: Density (g/cc) Vspheres Vcel % occupied Cubic (express to no. of atoms cm3 cm3 scientific Unit (whole no.) notation; round- (round-off to two | (round-off to two decimal places) Cell (round-off to two decimal places) decimal places) off to two decimal places) SC x10-24 FCC x10-24 ВСС x10-24 Based on the calculated density, what is the most probable lattice type of aluminum metal? Body Centered Cubic (BCC) Structure, Simple Cubic (SB) Structure, or Face-Centered Cubic (FCC) Structure?
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter9: Liquids, Solids, And Materials
Section: Chapter Questions
Problem 115QRT
Related questions
Question
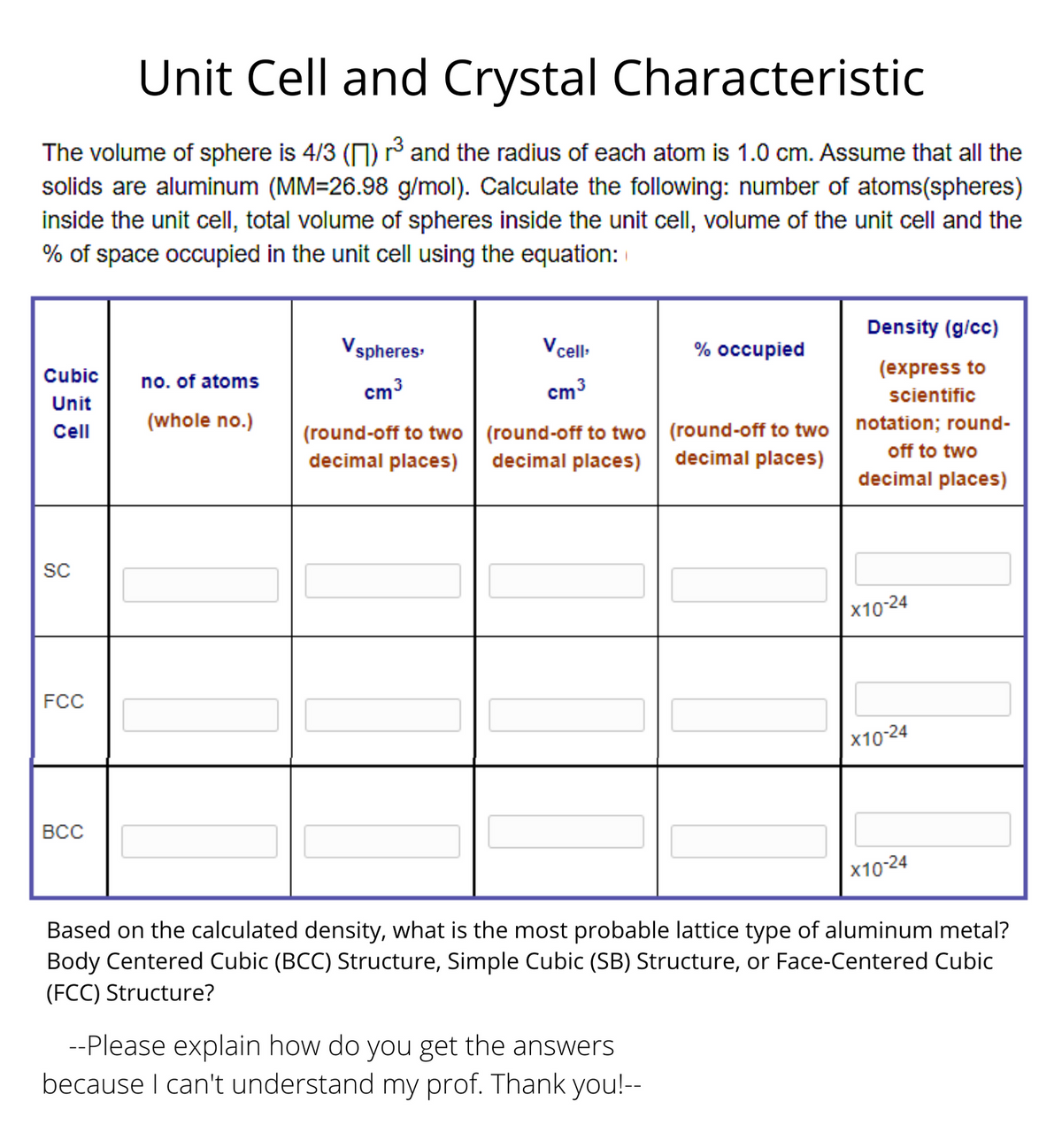
Transcribed Image Text:Unit Cell and Crystal Characteristic
The volume of sphere is 4/3 (1) ³ and the radius of each atom is 1.0 cm. Assume that all the
solids are aluminum (MM=26.98 g/mol). Calculate the following: number of atoms(spheres)
inside the unit cell, total volume of spheres inside the unit cell, volume of the unit cell and the
% of space occupied in the unit cell using the equation:
Density (g/cc)
Vspheres
V cel
% occupied
Cubic
(express to
no. of atoms
cm3
cm3
scientific
Unit
(round-off to two | (round-off to two | (round-off to two | notation; round-
decimal places) | decimal places)
(whole no.)
Cell
decimal places)
off to two
decimal places)
SC
X10-24
FCC
X10-24
ВсС
|x10-24
Based on the calculated density, what is the most probable lattice type of aluminum metal?
Body Centered Cubic (BCC) Structure, Simple Cubic (SB) Structure, or Face-Centered Cubic
(FCC) Structure?
--Please explain how do you get the answers
because I can't understand my prof. Thank you!--
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning