Chapter9: Complexometric And Precipitation Titrations
Section: Chapter Questions
Problem 3P
Related questions
Question

Transcribed Image Text:Data Report Sheet: Quantitative Analysis of Group I Cations
Observations on Standard Solution:
Part B:
Part C:
Part D:
Unknown
Unknown #
89
Redraw the flow chart for this experiment and record you observations for each step.
Ions Present in Unknown:
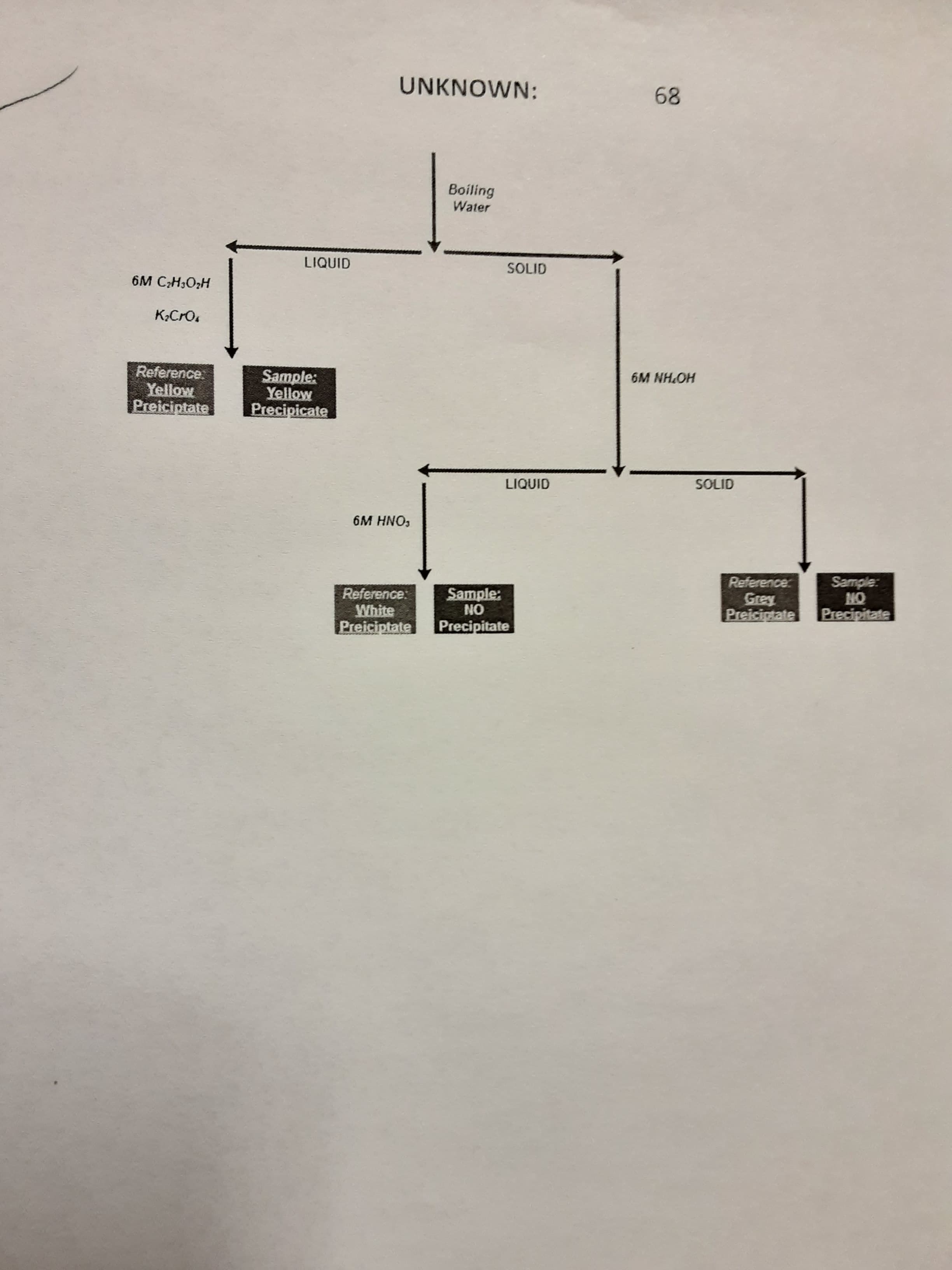
Transcribed Image Text:UNKNOWN:
89
Boiling
Water
LIQUID
SOLID
6M C;H,O;H
K,CrO,
6M NH.OH
Reference:
Yellow
Preiciptate
Sample:
Yellow
Precipicate
LIQUID
SOLID
6M HNO,
Reference:
Sample:
ON
Precipitate
Sample:
Reference:
White
Preiciptate
Preiciptate
ON
Precipitate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

