Unknown Mixture #1 Unknown Mixture #2 Sample caffeine ibuprofen aspirin acetaminoph en Distance to solvent front 14,70 cm 14,70cm 14.70cm 14.70cm1u.70 cm 14.70cm 5. 70cm 11.90cm I1.85cm Distance Spot(s) traveled 1.75 cm 13.75cm 11.9 5cm 6.40 cm 13. 75 cm 13,75 cm .38 78 . 8095 .8061 Calculated R(s) 119 9354 8129 43 5 9354 .9354 #1:Aspirin and lbuprofen Components of Unknowns #2: Ace taminophen, aspirin an d ibu profen D. Additional Exercises 1. Which of the substances tested is most polar? What parts of the structure of this substance is polar? The su 2. Which of the substance tested is most non-polar? What parts of the structure of this substance in non-polar? po the
Unknown Mixture #1 Unknown Mixture #2 Sample caffeine ibuprofen aspirin acetaminoph en Distance to solvent front 14,70 cm 14,70cm 14.70cm 14.70cm1u.70 cm 14.70cm 5. 70cm 11.90cm I1.85cm Distance Spot(s) traveled 1.75 cm 13.75cm 11.9 5cm 6.40 cm 13. 75 cm 13,75 cm .38 78 . 8095 .8061 Calculated R(s) 119 9354 8129 43 5 9354 .9354 #1:Aspirin and lbuprofen Components of Unknowns #2: Ace taminophen, aspirin an d ibu profen D. Additional Exercises 1. Which of the substances tested is most polar? What parts of the structure of this substance is polar? The su 2. Which of the substance tested is most non-polar? What parts of the structure of this substance in non-polar? po the
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter23: Polymeric Materials And Soft Condensed Matter
Section: Chapter Questions
Problem 12P
Related questions
Question
100%
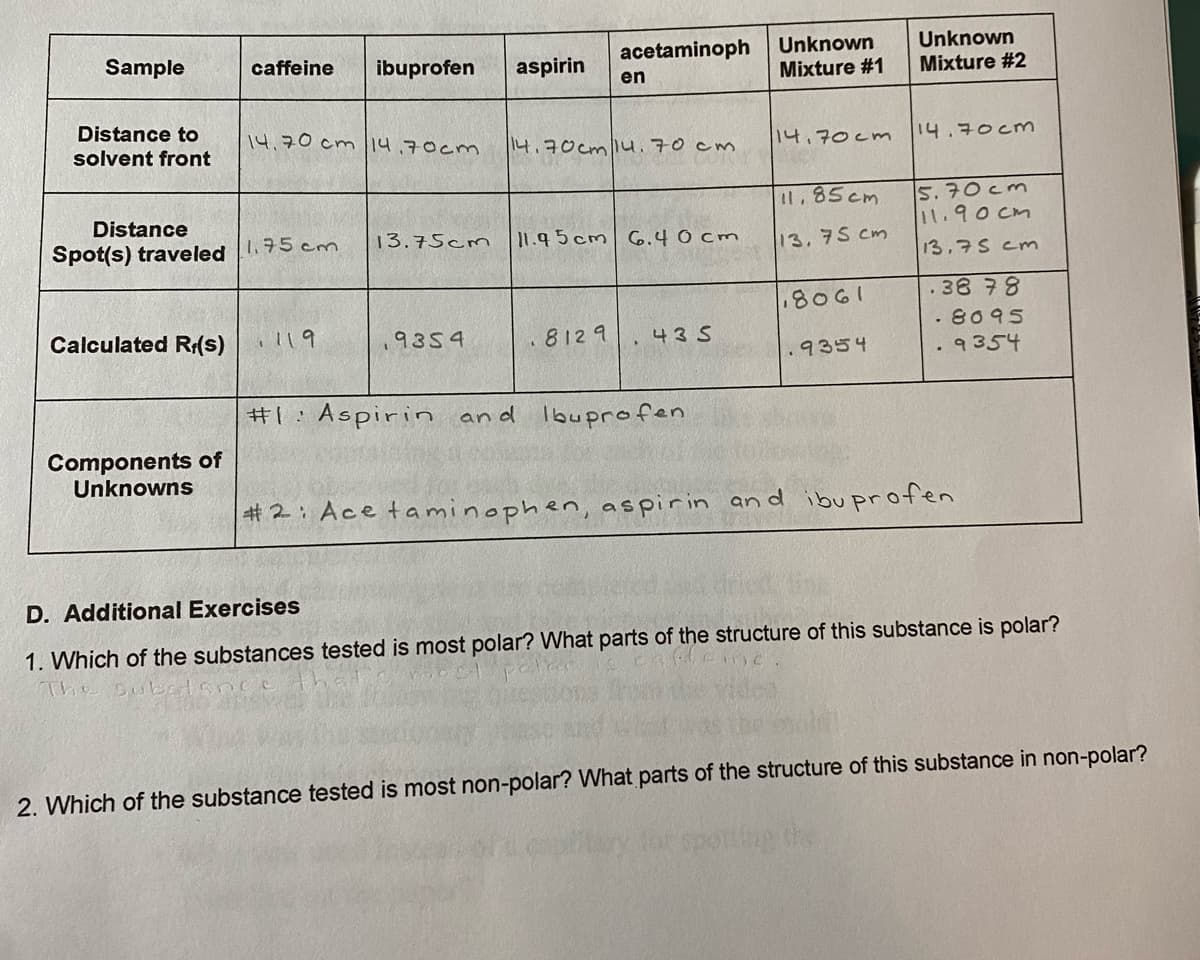
Transcribed Image Text:Unknown
Mixture #1
Unknown
Mixture #2
Sample
caffeine
ibuprofen
aspirin
acetaminoph
en
Distance to
solvent front
14,70 cm 14,70cm
14.70cm1u.70 cm
1나,구 cm
|14.구0cm
5. 70 cm
11.90 cm
I.85cm
Distance
Spot(s) traveled 175 cm
13.75cm
11.9 5 cm 6.4 0 cm
13,75 cm
13.75 cm
38 78
1908
.8095
Calculated R:(s)
119
,9354
812 9
43 S
.9354
9354
#1:Aspirin
an d lbuprofen
Components of
Unknowns
#2: Ace taminophen, aspirin
an d ibu profen
D. Additional Exercises
1. Which of the substances tested is most polar? What parts of the structure of this substance is polar?
The Sul
2. Which of the substance tested is most non-polar? What parts of the structure of this substance in non-polar?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax