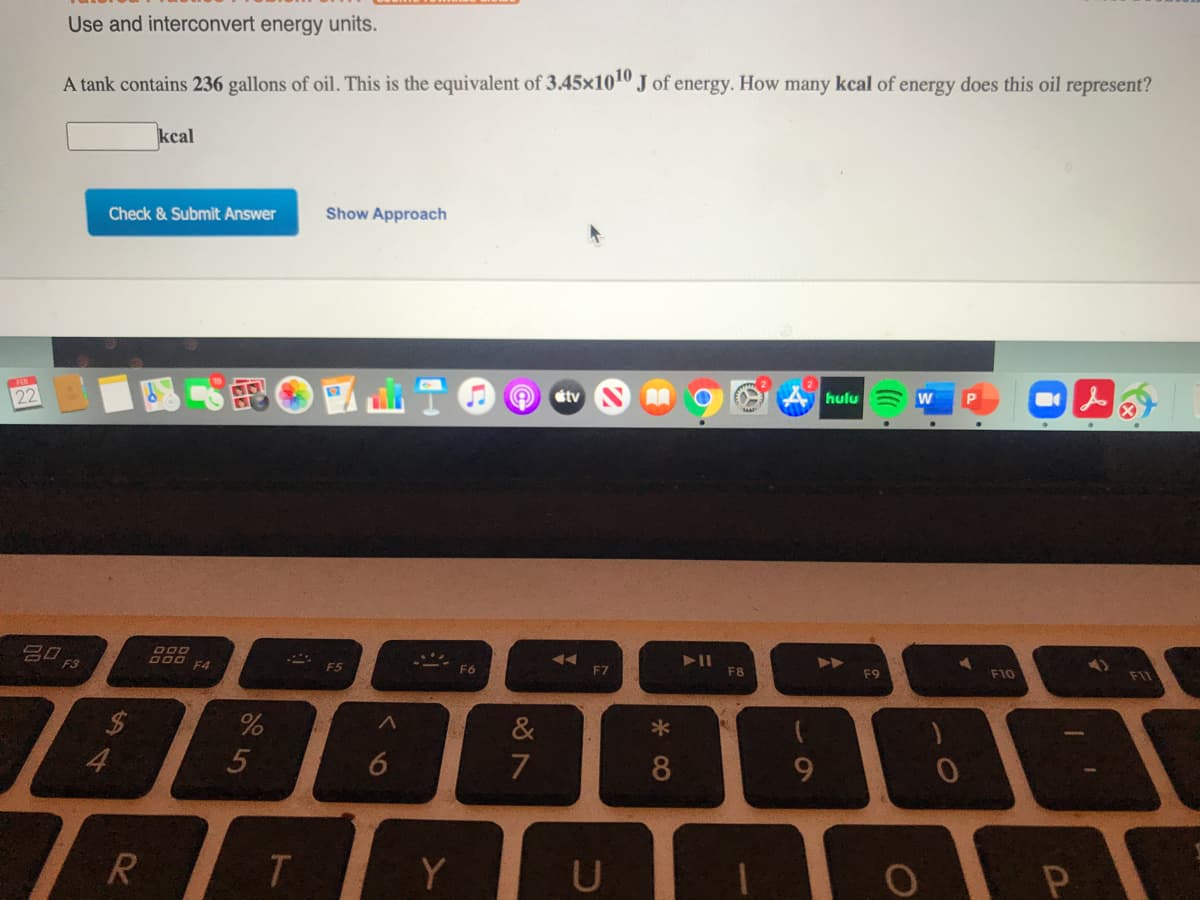
Use and interconvert energy units. A tank contains 236 gallons of oil. This is the equivalent of 3.45x100 J of energy. How many kcal of energy does this oil represent? kcal Check & Submit Answer Show Approach
Q: A typical person has an average heart rate of 69.0 beats/min. Calculate the given questions. How…
A: The number of significant figures in the product of the calculation is the minimum number of…
Q: note: just answer item 1
A:
Q: =1d3e42816b27b369fba593da0106e31d#10001 5 of 13 Review| Constants | Periodic Table Part B How much…
A: Generally, Number of atoms/Molecules = Number of moles x NA
Q: Listed below you have been given a focus question that must be answered using a well-rounded…
A: General chemistry
Q: [References] This question has multiple parts. Work all the parts to get the most points. a…
A: Answer: -390 KJ
Q: need step by need help with calculations! step answers please. acid is HCI, thank you :) Data Beaker…
A: Please find the attachment.
Q: Find calculated k value per trial: Average k value: The initial volume of syringe was 15ml and I…
A: To find calculated average k value per trial and average k value.
Q: I Review | Constants Periodic Table Part C Consider the following two expressions, the only…
A:
Q: older. Or import from another browser. Import favorites [Review Topics] [References] Some…
A: The amount of gasoline (V) is required to drive the car 9.00 miles is calculated as shown below…
Q: Answer is provided. All I need is the solution. Thank you! A patient cuts 100-mg scored tablets to…
A:
Q: Review Constants I Periodic Tab Part A One cup of clam chowder contains 21 g of carbohydrete, 11 g…
A: Given that 1 cup of Clam Chowder has 21 g carbohydrate, 11 g fat, and 9 g protein. Given the…
Q: the
A: According to the question, Katrina inserted a key into her car’s ignition, then she turns the key,…
Q: Part A Find A:U for the combustion of ethanol (C,H:OH) to carbon dioxide and liquid water from the…
A: The question is based on the concept of specific heat capacity. it is defined as the amount of…
Q: QUESTION 2 Consider the following data concerning the equation: H202+ 31+ 2H*- 13+2H20 [H2021 [H*)…
A: In this type of order determining method, rate is compared in two steps keeping all otger reactants…
Q: A paraffin wax candle is 6.25 inches tall and 1.50 inches in diameter. How many mL of paraffin wax…
A:
Q: +Table 2. Iron ore and cerium solution data. Flask Flask Flask 1 3 Iron ore mass, g: 0.105…
A: Given: The concentration of standard Ce(SO4)2 solution is 0.0362 M. The mass of iron ore taken in…
Q: Chrome File Edit View History BookmarkS People Window Help A 1 OF Chemistry - S2 - MI, T2 - Activ x…
A: Given, Volume = 8.0 L Pressure = 2.5 atm Moles of gas = 3 moles Temperature = ?
Q: Your doctor gives you a prescription. The dosage for the prescription is 5 mL for every 3 pounds of…
A: Given: According to the doctor prescription 5 ml for every 3 pounds Child weight=84 pounds To find:…
Q: Indicate whether the following experimental mistake will increase, decrease, or have no effect on…
A:
Q: A particular beer is 6.50 % ethanol by volume. If a single bottle of beer contains a total of 750.0…
A:
Q: ed Test This test has a time limit of 1 hour.This test will save and submit automatically when th…
A: Gibbs Free Energy - Gibbs Free Energy can be most appropriately can be defined as the useful work…
Q: 1. The fertilizer recommendation for a 1 hectare of rice farm during dry season is 120-60-60kg…
A: At First you have to calculate the amount of phosphorus and potassium fertilizers amount because…
Q: Exploring the propitiates of gases experiment. Please show calculations. Volume (mL) Pressure (atm)…
A:
Q: 125% v +. Zoom Add Page Insert Table Chart Text Shape Media Comment Consider the following image of…
A: The first image shows the buret initial reading at 11.2mL. The second image shows the final reading…
Q: Data of candy (trial 1) Mass of candy: 6.1115g Concentration of NaOH (M): 0.09639 Volume of NaOH…
A: Interpretation: We have to determine the volume of NaOH required at the end point .
Q: Please estimate the Km and Vmax and then calculate the kinetic constants (Km, Vmax, kcat, and…
A: Estimation of Km and Vmax can be done from the Michaelis-Menten equation. The values of Km and Vmax…
Q: Case study: Ms FP and some friends go to a restaurant for lunch. They each order the 'special of the…
A: Energy by per gram of component : Energy by one gram of carbohydrate = 4 kcal Energy by one gram of…
Q: Food safety guidelines recommend that a beef rib roast (bone-in) should cook for 23.0 minutes per…
A: The relations to put within the fractions are: 23.0 mins = 1 pound 1 hour = 60 mins
Q: E Chemistry Review - Google E Stoichiometry Lab b Answered: Reevaluate your x M Verify your email…
A:
Q: Pre what will be observed in each experiment below. predicted observation (choose one) experiment…
A: In order to understand and interpret the answer of the above situations we need to know the effect…
Q: On the average, during the Winter 2022 Olympics and training, a male cross-country skiing athlete…
A:
Q: Activity 1 Directions: Measure the ingredients of a recipe using the ordinary and standard measuring…
A: Most measuring tools use a ruler or a scale as standard units of measurement. Non-standard units of…
Q: Botulin toxin has an estimated LD50 value of 200 pg/kg of body weight and an estimated molar mass of…
A:
Q: What is the value of 252 calories in joules (J)? Type answer:
A:
Q: Calculate the % (m/m) of helium gas
A:
Q: This question was rejected, but this question is part of a practice set and as you can see it is out…
A: According to the ideal gas law PV = nRT where P = pressure of gas V = volume of vessel = 25.9 L…
Q: (Application) Bananas Foster is an example of a dessert that is flambéed. A Bananas Foster label…
A:
Q: Please analyze the given and answer the question number 1. Compute for the actual weight of each…
A: A question based on introduction to chemistry that is to be accomplished.
Q: While in Europe, if you drive 119 km per day, how much money would you spend on gas in one week if…
A: Given: Distance travelled in one day = 119 km Distance travelled in one day = 7×119 km = 833 km Cost…
Q: Your doctor prescribed eye drops for you to treat a certain condition. You obtain the eye drop…
A:
Q: Please choose the letter of the correct answer. I will rate your answer 3. Extra energy is…
A: The biological significance of organic compounds, its structure, reactivity, and interactions are…
Q: Convert -25OC to a temperature in K.
A:
Q: 2. Scatter or bar? 3. Scatter or bar? 100 100 Reason Reason * 75 75 50 50 25 Question 25 Question 4…
A: Scatter Graph - In this type of graph only points are drawn where our desired results are. We…
Q: Manipulate equation 3 to match the H20 part of the overall equation. What will be the new AH value?…
A:
Q: Determine the formal charge on each atom in the structure.
A: The structure given is,
Q: collect Data: ( ● Use the simulation to determine the final temperature for each set-up in the table…
A:
Q: Use and interconvert energy units. A tank contains 176 gallons of oil. This is the equivalent of…
A:
Q: There are 12 eggs in a dozen. If a farmer's chickens produce an average of 284 dozen eggs in a…
A:
Q: Select D if neither statement A nor B are true 1. Fertilizer X is a complete fertilizer with a…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The following reaction using hydrogen and oxygen is carried out in a bomb calorimeter: 2H2(g) + O2(g) → 2H2O(l) The specific heat of the reaction vessel is 0.200 kcal/0C-kg and the specific heat of water is 1.00 kcal/0C-kg. Assuming that m is 0.2 mole of water was formed in the experiment and Q is 4.4 kcal, calculate the heat of the reaction per mole of liquid water.The specific heat of an unknown liquid is determined using a sample of molybdenum (Mo) with a mass of 35.12g. The Mo is heated to 99.2OC in boiling water, and then transferred to 52.16g of unknown liquid whose temperature is 23.4OC. The final temperature of the Mo-unknown liquid system is 28.9OC. Calculate the specific heat of the unknown liquid. CMo = 0.251 J•g-1•OC-1Use the molar DHf° under the formulas to calculate DH°rxn for the equations as balanced: 1. 2B2H6(g) + 3CO2(g) --> 2B2O3(s) + 3CH4(g) ΔH°rxn = _______ kJ ΔH°f = +36 –394 –1274 –75 kJ/mol exo ? endo_thermic 2. 2P2O5 + 2CaC2 -->P4 + 2CaCO3 + 2CO2 ΔH°rxn = _______ kJ ΔH°f = –1505 –59 ___ –1207 –394 kJ/mol exo ? endo_thermic 3. 2Na2CrO4(s) + 10HCl(g) --> 4NaCl(s) + 3Cl2(g) + Cr2O3(s) + 5H2O(l) ΔH°f = –1342 –92 –411 ___ –1140 –286 kJ/mol ΔH°rxn = _______ kJ exo ? endo_thermic
- The density of all solutions are 1.0g/mL and their respective heat capacities 4.184J/g.K. NH4OH(aq)+ HCl(aq)--> NH4Cl(aq)+H2O(l) Volume of 2.0 M HCl 25.0mL NUmber of moles of HCl ? Temperature of HCl 15oC Volume of 2.0 M NH4OH 25.0 mL Number of moles of NH4OH ? Temperature of NH4OH 15oC Intitial Temperature 15oC Final Temperature 23.5oC Change in Temperature ? Calculate qr in J (qr=-qsol) using qr=-mC(change in temperature). What is the enthalpy change ( Delta H) in kJ.mol-1, when 1 mole of acid is neutralized? delta H=qr/nHClA 3kg cat is placed in a calorimeter with 1000mls of water being circulated through coils at the top of thechamber for 2 hours. During that time the calculated change in water temperature is 13°C.a. What is the metabolic rate of the cat in both kcal/hr as well as kJ/hr?b. What would the daily energy requirements be for the cat?c. What is the cat’s metabolic rate per kg of body mass?A pellet of benzoic acid standard was combusted in a bomb calorimeter to determine the experimental calorimeter constant. After 6 mins of monitoring the temperature, the bomb was fired. The following data were obtained. mole of benzoic acid: 0.0049 mol mass of burnt fuse wire: 0.0125 g qwire,surr: 5858 J/g ΔHcombustion of benzoic acid: -3228.0 kJ/mol Δng (combustion of benzoic acid): -0.5 mol R: 8.314 J/mol•K r1: 0ºC/min r2: 0ºC/min Tf: 30.4ºC Ti: 28.4ºC Determine the calorimeter constant (in J/ºC). Report your answer in 2 decimal places.
- One kilogram of boiling water is poured into an empty thermos whose mass is 1kg and whose temperature is 25oC. No heat is lost from the thermos. The specific heats of water and the thermos are 1 and 0.5 kcal/kg-oC, respectively. Their final temperature is?In constant pressure calorimeter 75.0 ml is mixed with 1.25 M Hydrocloric acid solution is mixed with 75.0 ml of 1.25 M Sodium hydroxide solution. the density of the final solution is 1.00g/ml and the the solution both initially 21.45c, reach a maximum temperature of 28.32c when mixed. based on this information, and estimating the solution's heat capacity as 4.18Jg-1c-1, what is the amount of heat in Kilojoules, transferred in this reaction?A 75.0 mL. of 2.00 MHCl(aq) at 22.0 oC is added to 150.0 mL. of 0.300 M NaOH(aq) at 22.000 oC. Then the reaction is proceeded and the final temperature is reached to 28.000 oC. Calculate the heat of reaction in kJ / mole. Hint: Treat this mixture as a diluted solution. CP of H2O(l) = 4.184 J / g oC D of H2O = 0.988 g/ mL. a. -37.3 kJ / moleb. -55.9 kJ/ molec. 124 kJ / moled. -124 kJ / mole
- Mass of Empty Calorimeter is 7.73g, mass of calorimeter + water is 46.875g, mass of water is 39.145g, mass of metal is 6.354g, initial temperature of water is 20 degrees, initial temperature of metal is 100 degrees Celsius, Final temperature of calorimeter is 21.5 degrees Celsius, T(T-Final - T-Initial) is 78.5 degrees Celsius, q-calorimeter is 1841.29J, Specific Heat c, of Metal is 3.69 J/g degrees Celsius, Calculate molar mass of metal.Using data for DHof (KJ/mol) provided below, calculate DHoRxn for: N2O(g) + 3H2(g) ---> N2H4 (l) + H2O (l) DHof (KJ/mol): N2O(g) = 33.85 ; H2 (g)= 0 ; N2H4 (l)= 50.4 ; H2O(l) = -285.8To determine the specific heat of a newly created, highly conducting material, a 704.8 mg sample of the material was supplied with 35.548 J of heat causing the temperature of the material to rise from 297.05 K to 461.4 K. What is the specific heat of this material? Report your answer in units of J/(ºC · g) to 4 sig figs, but do NOT include units in your answer.

