Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas that should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume that the water is initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a nitrogen partial pressure of 0.78 atm. Express your answer using two significant figures. Π| ΑΣΦ Vtotal = 0.22 Submit Previous Answers Request Answer www. ? L
Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas that should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume that the water is initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a nitrogen partial pressure of 0.78 atm. Express your answer using two significant figures. Π| ΑΣΦ Vtotal = 0.22 Submit Previous Answers Request Answer www. ? L
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 66QAP: The Henry's law constant for the solubility of radon in water at is 9.57106 M/mm Hg. Radon is...
Related questions
Question
The small bubbles that form on the bottom of a water pot that is being heated (before boiling) are due to dissolved air coming out of solution.
Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas that should bubble out of 1.2 L of water upon warming from 25 C to 50 °C. Assume that the water is initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a nitrogen partial pressure of 0.78 atm
Express your answer using two
Transcribed Image Text:The small bubbles that form on the bottom of a
water pot that is being heated (before boiling) are
due to dissolved air coming out of solution.
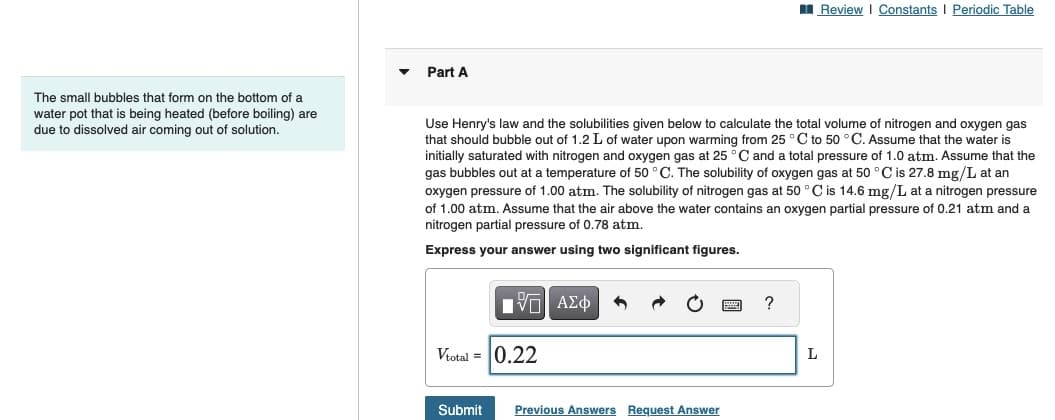
Part A
Use Henry's law and the solubilities given below to calculate the total volume of nitrogen and oxygen gas
that should bubble out of 1.2 L of water upon warming from 25 °C to 50 °C. Assume that the water is
initially saturated with nitrogen and oxygen gas at 25 °C and a total pressure of 1.0 atm. Assume that the
gas bubbles out at a temperature of 50 °C. The solubility of oxygen gas at 50 °C is 27.8 mg/L at an
oxygen pressure of 1.00 atm. The solubility of nitrogen gas at 50 °C is 14.6 mg/L at a nitrogen pressure
of 1.00 atm. Assume that the air above the water contains an oxygen partial pressure of 0.21 atm and a
nitrogen partial pressure of 0.78 atm.
Express your answer using two significant figures.
IVE ΑΣΦ
Vtotal = 0.22
Submit
Previous Answers Request Answer
?
Review | Constants | Periodic Table
L
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning