Use stoichiometric methods to analyze a mixture. A mixture of MgCl, and inert material is analyzed to determine the Mg content. First the mixture is dissolved in water. Then all of the chloride ion is precipitated as AgCl by the addition of an excess of silver nitrate. MgCly(aq) + 2A£NO3(aq) 2A£C(s) + Mg(NO3)½(aq) In one experiment, a 0.5770 g sample of the mixture resulted in 1.3717 g of AgCl. Determine the percent (by mass) of Mg in the mixture. % Mg
Use stoichiometric methods to analyze a mixture. A mixture of MgCl, and inert material is analyzed to determine the Mg content. First the mixture is dissolved in water. Then all of the chloride ion is precipitated as AgCl by the addition of an excess of silver nitrate. MgCly(aq) + 2A£NO3(aq) 2A£C(s) + Mg(NO3)½(aq) In one experiment, a 0.5770 g sample of the mixture resulted in 1.3717 g of AgCl. Determine the percent (by mass) of Mg in the mixture. % Mg
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter6: Types Of Chemical Reactions And Solution Stoichiometry
Section: Chapter Questions
Problem 126CP
Related questions
Question
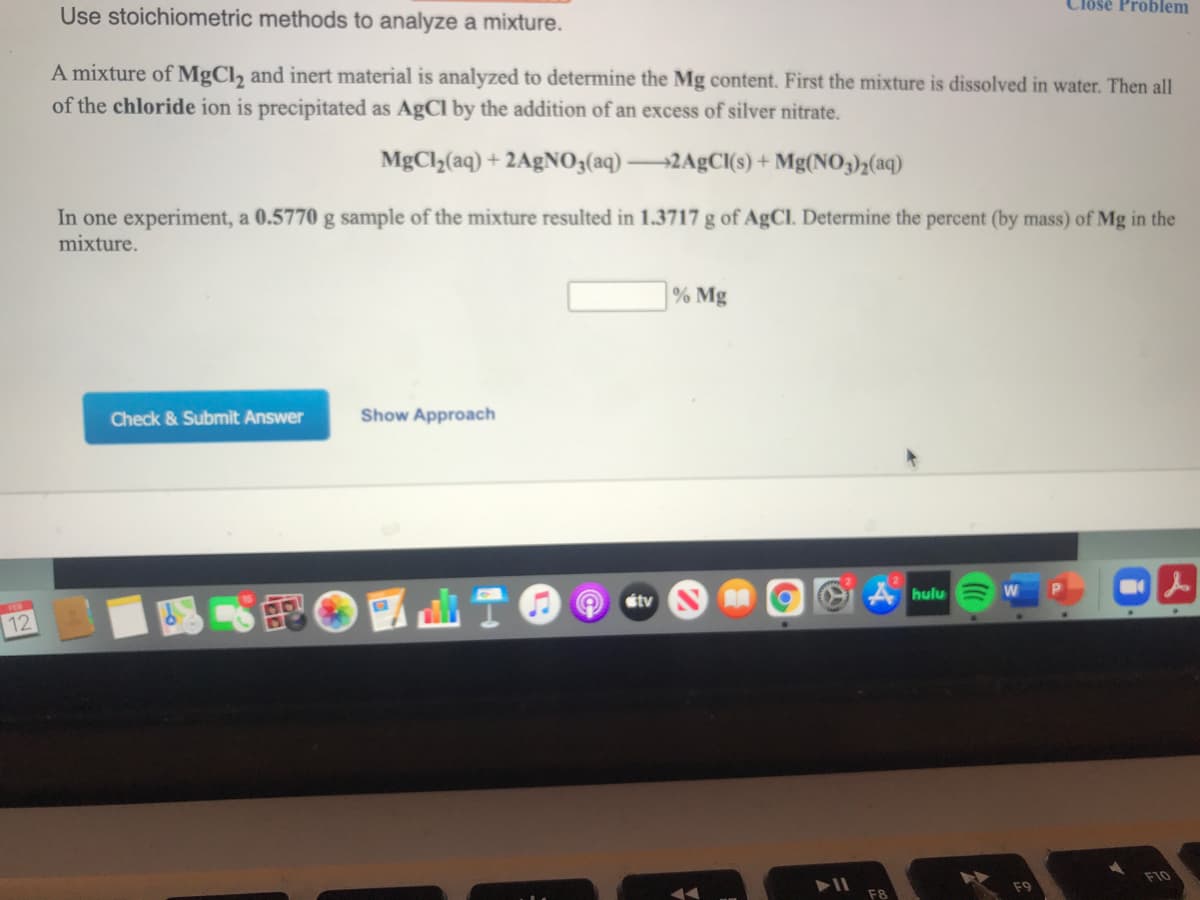
Transcribed Image Text:Use stoichiometric methods to analyze a mixture.
Close Problem
A mixture of MgCl, and inert material is analyzed to determine the Mg content. First the mixture is dissolved in water. Then all
of the chloride ion is precipitated as AgCl by the addition of an excess of silver nitrate.
MgCl2(aq) + 2A£NO3(aq) 2A£CI(s) + Mg(NO3)½(aq)
In one experiment, a 0.5770 g sample of the mixture resulted in 1.3717 g of AgCl. Determine the percent (by mass) of Mg in the
mixture.
% Mg
Check & Submit Answer
Show Approach
A hulu
FEB
étv
12
F10
F8
Expert Solution
Step 1
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning