Use the data given below to calculate the bond energies (Do) (in eV) of 'H3³CI and 'H®lBr molecules. Which bond is stronger? Molecule v. (cm¯1) D.((cm¯1) HCI 2991 53194 HBr 2649 34570
Use the data given below to calculate the bond energies (Do) (in eV) of 'H3³CI and 'H®lBr molecules. Which bond is stronger? Molecule v. (cm¯1) D.((cm¯1) HCI 2991 53194 HBr 2649 34570
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 38P: The percent ionic character of the bonds in several interhalogen Molecules (as estimated from their...
Related questions
Question
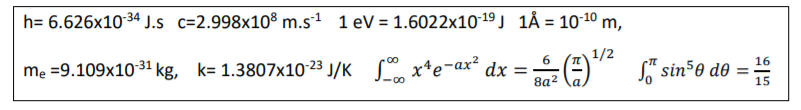
Transcribed Image Text:h= 6.626x10 34 J.s c=2.998x10% m.s1 1 eV = 1.6022x10-19 J 1Å = 1010 m,
%3D
6.
1/2
mẹ =9.109x1031 kg, k= 1.3807x10-23 J/K Sº x*e¬ax² dx =-
8a2
16
S" sin50 de =-
15

Transcribed Image Text:Use the data given below to calculate the bond energies (Do) (in eV) of
'H³$Cl and 'H8' Br molecules. Which bond is stronger?
Molecule ve (cm-1) De((cm-1)
HCI
2991
53194
HBr
2649
34570
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning