Use the data in the graph above to estimate a KM value for the enzyme in the presence of these metabolites, and enter them into the table below. b. Classify these metabolites as either activators or inhibitors, and explain your rationale below.
Use the data in the graph above to estimate a KM value for the enzyme in the presence of these metabolites, and enter them into the table below. b. Classify these metabolites as either activators or inhibitors, and explain your rationale below.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter25: Nitrogen Acquisition And Amino Acid Metabolism
Section: Chapter Questions
Problem 16P: A Deficiency on 3-Phosphogtycerate Dehydrogenase Can Affect Amino Acid Metabolism Although serine is...
Related questions
Question
4. a. Use the data in the graph above to estimate a KM value for the enzyme in the presence of these metabolites, and enter them into the table below.
b. Classify these metabolites as either activators or inhibitors, and explain your rationale below.
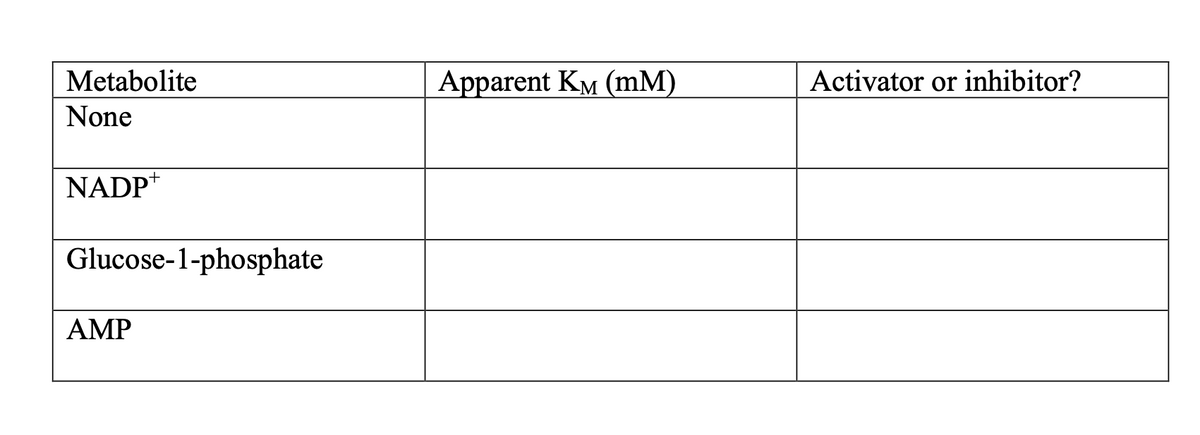
Transcribed Image Text:Metabolite
None
NADP+
Glucose-1-phosphate
AMP
Apparent KM (mm)
Activator or inhibitor?
![The activity of the GAPDH
enzyme was assayed in the
presence of a constant
amount of glyceraldehyde-
3-phosphate and an
increasing amount of
NAD. The activity of the
control (that is, the enzyme
with no other metabolites)
was compared to the
activity of the enzyme in
the presence of various
metabolites. The results are
shown in the figure to the
right.
Units/mg protein
R
8
9
10
-NADP+
Hill coeficient = 1
Hill coefficient = 2
NAD], mM
15
Control -- AMP-X-Glucose-1-P
20](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F0627210d-c0e3-4a2a-8541-a7399a2e5e96%2Ff328a2bf-debe-40e7-8c5d-a7c75899496b%2Fwnuzmos_processed.png&w=3840&q=75)
Transcribed Image Text:The activity of the GAPDH
enzyme was assayed in the
presence of a constant
amount of glyceraldehyde-
3-phosphate and an
increasing amount of
NAD. The activity of the
control (that is, the enzyme
with no other metabolites)
was compared to the
activity of the enzyme in
the presence of various
metabolites. The results are
shown in the figure to the
right.
Units/mg protein
R
8
9
10
-NADP+
Hill coeficient = 1
Hill coefficient = 2
NAD], mM
15
Control -- AMP-X-Glucose-1-P
20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning