Use the dilution equation to calculate the starting concentration of each reactant for each trial. (Hint: You know the concentration and volume of each reactant added, M1 and V1. For every trial, the total volume, V2, is 1.9 mL.)
Use the dilution equation to calculate the starting concentration of each reactant for each trial. (Hint: You know the concentration and volume of each reactant added, M1 and V1. For every trial, the total volume, V2, is 1.9 mL.)
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter14: Chemical Kinetics: The Rates Of Chemical Reactions
Section: Chapter Questions
Problem 85IL: The color change accompanying the reaction of phenolphthalein with strong base is illustrated below....
Related questions
Question
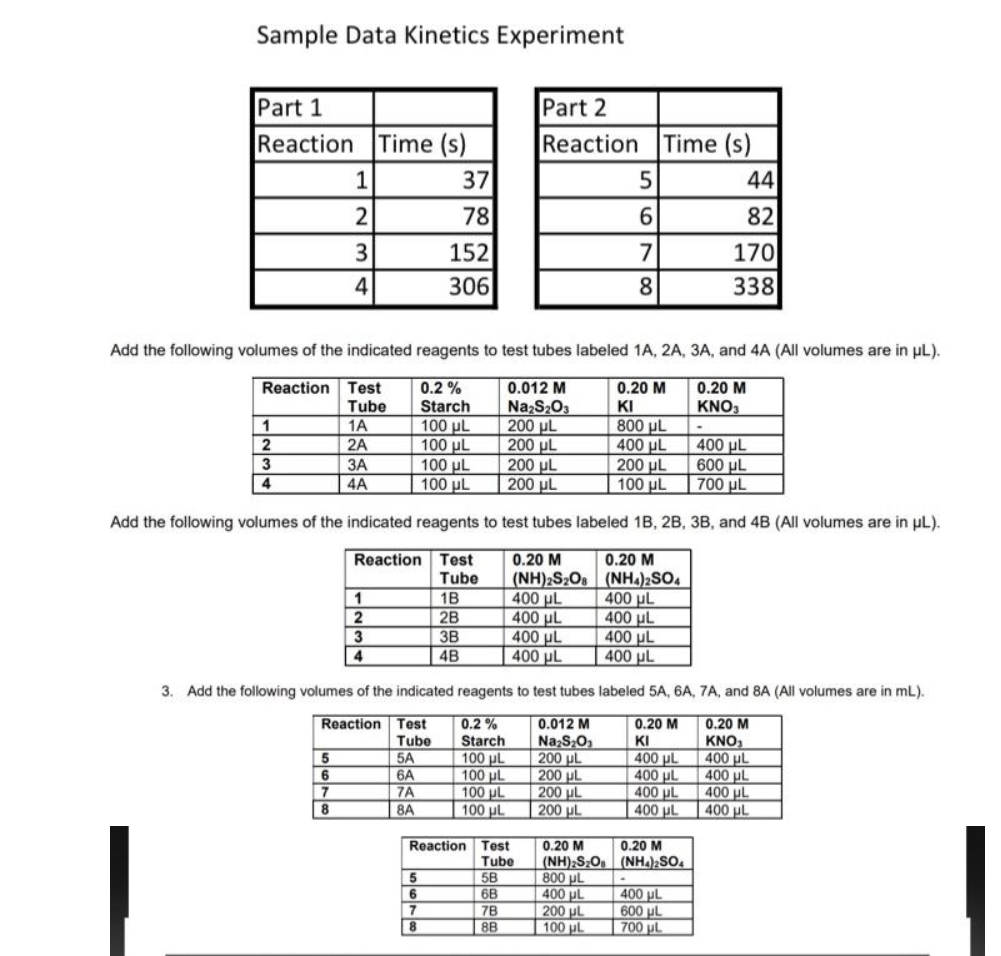
For parts 1 and 2, Use the dilution equation to calculate the starting concentration of each
reactant for each trial. (Hint: You know the concentration and volume of each reactant added,
M1 and V1. For every trial, the total volume, V2, is 1.9 mL.)
Kinetics Data
| Part 1 | Part 2 | ||
| Reaction | Time (s) |
Reaction |
Time (s) |
| 1 | 37 | 5 | 44 |
| 2 | 78 | 6 | 82 |
| 3 | 152 | 7 | 170 |
| 4 | 306 | 8 | 338 |
0.2% starch, 0.012M Na2S2O3, 0.20M KI, 0.20M KNO3, 0.20M (NH4)2S2O8, 0.20M (NH4)2SO4, 0.0020M Cu(NO3)2
Transcribed Image Text:Sample Data Kinetics Experiment
Part 1
Part 2
Reaction Time (s)
Reaction Time (s)
1
37
44
2
78
82
3
152
7
170
4
306
8.
338
Add the following volumes of the indicated reagents to test tubes labeled 1A, 2A, 3A, and 4A (All volumes are in uL).
Reaction Test
Tube
1A
0.2 %
Starch
0.012 M
0.20 M
0.20 M
NazS2O3
200 uL
200 uL
200 µL
200 µL
KI
KNO,
1
2
100 pL
100 µL
100 µL
100 pL
800 pL
400 µL
200 µL
100 µL
400 uL
600 µL
700 pL
2A
3
ЗА
4
4A
Add the following volumes of the indicated reagents to test tubes labeled 1B, 2B, 3B, and 4B (All volumes are in uL).
Reaction Test
Tube
0.20 M
(NH)2S2Os (NH.)2SO4
400 µL
400 µL
400 pL
400 µL
0.20 M
400 uL
400 µL
400 µL
400 µL
1
1B
2B
3
3B
4B
3. Add the following volumes of the indicated reagents to test tubes labeled 5A, 6A, 7A, and 8A (All volumes are in mL).
Reaction Test
0.2 %
0.012 M
Na:S;O
200 ul
200 uL
200 uL
200 µL
0.20 M
0.20 M
Starch
100 µL
100 pL
100 pL
100 uL
KNO,
400 pL
400 ul
400 pL
400 uL
Tube
5A
KI
5
400 pL
6A
7A
400 µL
400 µL
400 uL
7
8
8A
0.20 M
(NH);S;O, (NH.)½SO4
800 µL
400 pL
200 uL
100 pL
Reaction Test
0.20 M
Tube
5
5B
6B
7B
8B
400 pL
|600 uL
700 pl
6
7
8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning