Use the following information to calculate the standard free energy change (AG°) and equilibrium constant (K) for the following system: CaCO3(s) = CaO(s) + CO2(g) T= 900.0 °C AH;° values (kJ/mol): CaO(s) = -634.9, CO2(g) = -393.51, CaCO3(s) =–1220.0 %3D S° values (J/mol K): CaO(s) = 110.0, CO2(g) = 213.8, CACO3(s) = 38.1 %3D %3D %3D
Use the following information to calculate the standard free energy change (AG°) and equilibrium constant (K) for the following system: CaCO3(s) = CaO(s) + CO2(g) T= 900.0 °C AH;° values (kJ/mol): CaO(s) = -634.9, CO2(g) = -393.51, CaCO3(s) =–1220.0 %3D S° values (J/mol K): CaO(s) = 110.0, CO2(g) = 213.8, CACO3(s) = 38.1 %3D %3D %3D
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 15P
Related questions
Question
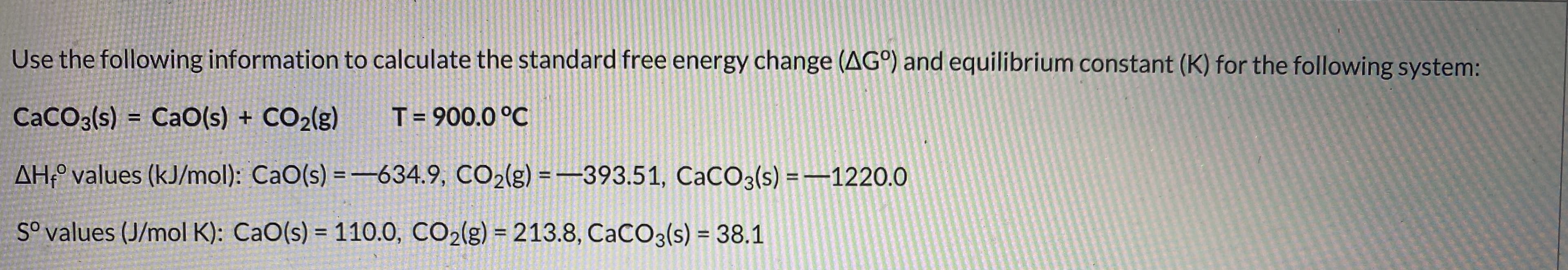
Transcribed Image Text:Use the following information to calculate the standard free energy change (AG°) and equilibrium constant (K) for the following system:
CaCO3(s) = CaO(s) + CO2(g)
T= 900.0 °C
AH;° values (kJ/mol): CaO(s) = -634.9, CO2(g) = -393.51, CaCO3(s) =–1220.0
%3D
S° values (J/mol K): CaO(s) = 110.0, CO2(g) = 213.8, CACO3(s) = 38.1
%3D
%3D
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning