Use the solubility table below to determine which of the following compounds is soluble? Solubility Rules Solubility Exceptions Solubility Exceptions carbonates insoluble Li, Na', K' , Rb', Cs*, NH, * insoluble Li", Na", K* , Rb*, Cs*, NH, * hydroxides insoluble Li', Na', K', Rb', Cs', NH, ', Ca²', Ba insoluble Li, Na', K* , Rb*, Cs*, NH, *, Mg*, Ca lon lon nitrates soluble none Ca" ,Ba" ,Sr2" , Ag* , Pb²* Ag', Pb sulfates soluble phosphates chlorides soluble alkali ions soluble none sulfides acetates soluble none soluble Ag*, Pb²* iodides О а. Ваз (РОд)2 O b. Fe(OH)3 O c. A93PO4 O d. A9C2H3O2 e. AgOH
Use the solubility table below to determine which of the following compounds is soluble? Solubility Rules Solubility Exceptions Solubility Exceptions carbonates insoluble Li, Na', K' , Rb', Cs*, NH, * insoluble Li", Na", K* , Rb*, Cs*, NH, * hydroxides insoluble Li', Na', K', Rb', Cs', NH, ', Ca²', Ba insoluble Li, Na', K* , Rb*, Cs*, NH, *, Mg*, Ca lon lon nitrates soluble none Ca" ,Ba" ,Sr2" , Ag* , Pb²* Ag', Pb sulfates soluble phosphates chlorides soluble alkali ions soluble none sulfides acetates soluble none soluble Ag*, Pb²* iodides О а. Ваз (РОд)2 O b. Fe(OH)3 O c. A93PO4 O d. A9C2H3O2 e. AgOH
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter5: Principles Of Chemical Reactivity: Energy And Chemical Reactions
Section5.8: Product- Or Reactant-favored Reactions And Thermodynamics
Problem 2.1ACP
Related questions
Question
Explain please

Transcribed Image Text:sility table below to determine which of the following compounds
Solubility Rules
olubility Exceptions
Solubility Exceptions
carbonates insoluble Li', Na', K' , Rb', Cs', NH, *
insoluble Li*, Na*, K* , Rb*, Cs*, NH, *
hydroxides insoluble Li, Na', K' , Rb', Cs', NH, *, Ca²", Ba²", Sr²*
lon
pluble
none
Ca" ,Ba" ,Sr²* , Ag* , Pb²*
Ag', Pb²"
pluble
phosphates
pluble
pluble
none
sulfides
insoluble Li, Na", K* , Rb°, Cs*, NH,*, Mg*, Ca", Ba", Sr²*
pluble
none
2+
pluble
Ag* , Pb²*
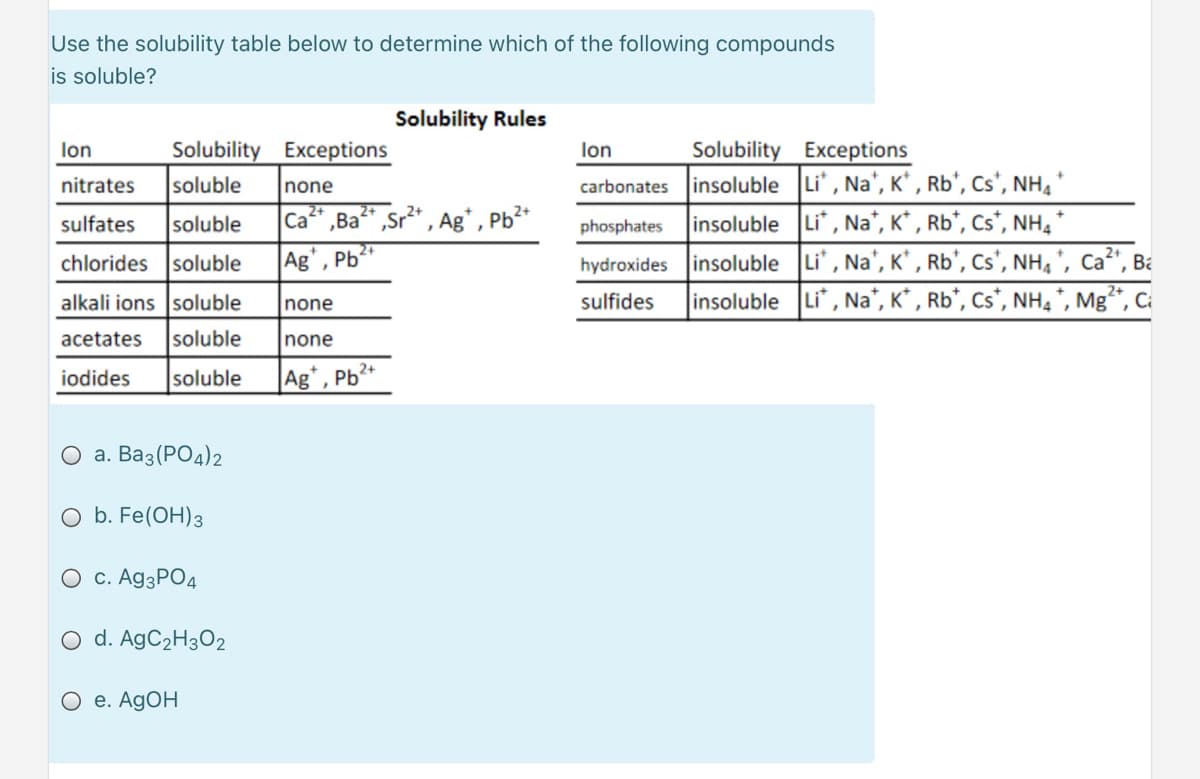
Transcribed Image Text:Use the solubility table below to determine which of the following compounds
is soluble?
Solubility Rules
Solubility Exceptions
Solubility Exceptions
carbonates insoluble Li, Na', K' , Rb', Cs', NH, *
insoluble Li", Na", K* , Rb*, Cs*, NH, *
hydroxides insoluble Li', Na', K' , Rb', Cs', NH, ', Ca²', Ba
insoluble Li, Na', K* , Rb*, Cs*, NH, *, Mg**, Ca
lon
lon
nitrates
soluble
none
Ca" ,Ba" ,Sr2* , Ag* , Pb²*
Ag', Pb
sulfates
soluble
phosphates
chlorides soluble
alkali ions soluble
none
sulfides
acetates
soluble
none
iodides
soluble
Ag*, Pb²*
О а. Ваз (РОд)2
O b. Fe(OH)3
O c. A93PO4
O d. AgC2H3O2
е. AgOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning