Using bond enthalpies, estimate the standard heat of the reaction for the drobromination of ethylene: Br H CH,=CH, + HBr ČH,-CH, TABLE 4-5. EMPIRICAL BOND ENTHALPIES AT 25°C (kcal/mole) Bond AH Bond ΔΗ C-CI H-H H-F 104 79 C-Br C-S 135 66 H-CI H-Br 0-0 0=0 0-H 103 62 C=8 C-N C=N 88 114 70 147 210 38 33 118 C=N N-N 111 C-H 99 C-0 N=N 100 84 170 C=0 C-C C=C C=C C-F N=N 226 N-H F-F 83 93 37 58 147 194 105 C-CI Br-Br C(s, graphite) - C(g) 172 46
Using bond enthalpies, estimate the standard heat of the reaction for the drobromination of ethylene: Br H CH,=CH, + HBr ČH,-CH, TABLE 4-5. EMPIRICAL BOND ENTHALPIES AT 25°C (kcal/mole) Bond AH Bond ΔΗ C-CI H-H H-F 104 79 C-Br C-S 135 66 H-CI H-Br 0-0 0=0 0-H 103 62 C=8 C-N C=N 88 114 70 147 210 38 33 118 C=N N-N 111 C-H 99 C-0 N=N 100 84 170 C=0 C-C C=C C=C C-F N=N 226 N-H F-F 83 93 37 58 147 194 105 C-CI Br-Br C(s, graphite) - C(g) 172 46
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter5: Principles Of Chemical Reactivity: Energy And Chemical Reactions
Section5.8: Product- Or Reactant-favored Reactions And Thermodynamics
Problem 1.2ACP
Related questions
Question
Please answer in 4 decimal places. Thank you!
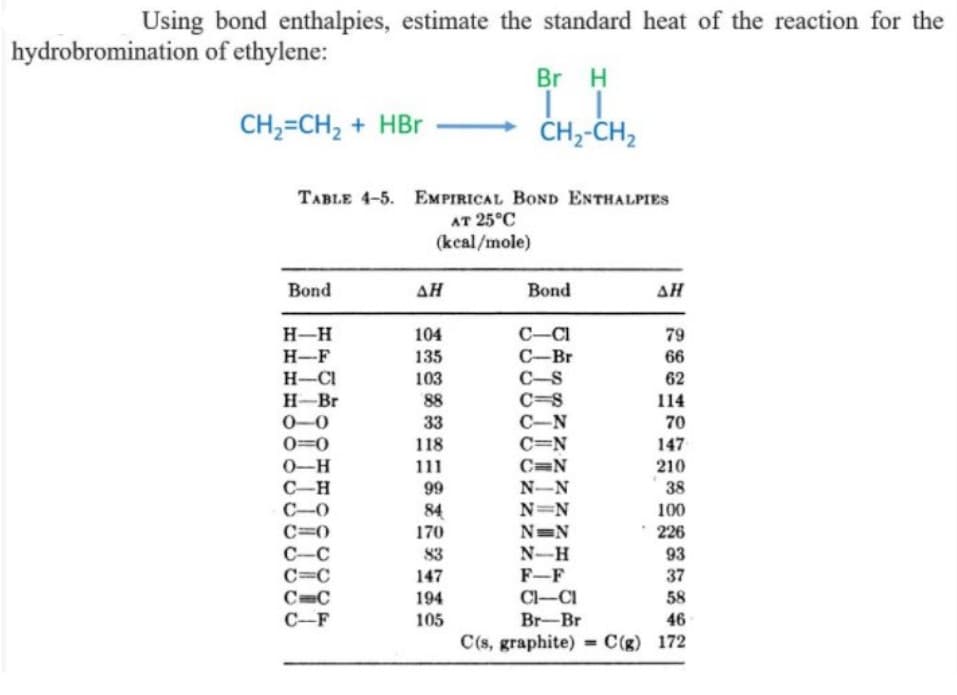
Transcribed Image Text:Using bond enthalpies, estimate the standard heat of the reaction for the
hydrobromination of ethylene:
Br H
CH,=CH, + HBr
ČH--CH,
TABLE 4-5. EMPIRICAL BOND ENTHALPIES
AT 25°C
(kcal/mole)
Bond
AH
Bond
ΔΗ
H-H
104
C-CI
79
H-F
135
C-Br
66
H-CI
H-Br
103
88
33
C-S
C=S
62
114
70
C-N
C=N
0-0
0=0
118
111
99
147
0-H
C=N
210
N-N
38
C-H
C-0
C=0
C-C
C=C
C=C
C-F
N=N
100
84
170
* 226
N=N
N-H
F-F
Cl-CI
Br-Br
83
93
37
147
194
58
105
46
C(s, graphite) =C(g) 172
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning