Q: Acetylsalicylic acid (C9H8O4) is a monoprotic acid commonly known as "aspirin." A typical aspirin ta...
A: Find out moles of NaOH and then use mol to mol ratio to find moles of aspirin. Convert that moles in...
Q: What is the molality of a solution made by dissolving 92g ethanol (C2H6O) in 200g water?
A:
Q: The solubility of silver nitrite, in water is 3.8 g/L. Calculate Ksp.
A: Given Solubility of AgNO3 (S) is 3.8 g/L first convert this into mol/ L S = 3.8 g / L x 1 / GM...
Q: A mixture of krypton and argon gas is compressed from a volume of 73.0 L to a volume of 30.0 L, whil...
A:
Q: Magnitude of the Equilibrium Constant 7 of 22 I Review | Constants | Periodic Ta Part D For a reacti...
A: k = (k forward)/(k backward)Since, k >1 => k(forward)> (k backward)=> equilibrium lies t...
Q: The element chlorine would be expected to form covalent bond(s) in order to obey the octet rule. Use...
A: The electronic configuration of H is 1s1.It means it has only one electron .so it will loose 1 elec...
Q: Find the molarity of Ba(OH)2 used by a laboratory analysts for standardization. It took 23.28 mL of ...
A:
Q: plant manufactures a high-grade phosphoric acid used in different laboratory experiments. The proces...
A:
Q: There is 0.25 mole of CO2 in a bottle of soda. How many grams of CO2 are in the bottle? 2. What coe...
A: We know mole is equal to mass divided by molar mass of given compound. So, by just putting the valu...
Q: A 12.63 g sample of calcium ore was dissolved in HCI and gravimetrically analyzed, through the preci...
A: Answer: These question are based on the stoichiometric calculation, where by calculation the moles o...
Q: Calculate the pH during the titration of 20.00 mL of 0.1000 M ethylamine, C2H5NH2(aq), with 0.1000 M...
A:
Q: Calculate the pH of a NHA*/NH3 buffer that was prepared with 100.0 mL of 0.500M NH,CI and 50.0 mL of...
A:
Q: Hydrogen reacts with oxygen to produce water as follows: 2H2(g) + O2(g) → 2H2O(g) How many moles ...
A: The reaction is as:- 2H2 + O2 ---> 2H2O
Q: For a 0.0494 M aqueous solution of cyanic acid, HCNO (Ka = 3.5*10-4), determine the following. What...
A: Given Data : 1. Molarity of cyanic acid solution = 0.0490M 2. Dissociation constant of cyanic acid K...
Q: Dissolving 3.0g of CaCl2 in 150.0g of water in a calorimeter at 22.4oC causes the temperature to ris...
A: Given, Mass of CaCl2 = 3 g Molar mass of CaCl2 = 110.98 g/mol Mass of Water, m = 150 g Initial te...
Q: If the [H3O+] is 4.95 x 10–12 M, what is the pH?
A: Given :- [H3O+] = 4.95 x 10–12 M To calculate :- pH of solution
Q: Part A Calculate A,G° for a redox reaction with n = 2 that has an equilibrium constant of K = 24 (at...
A: The Gibbs free energy change and the equilibrium constant are related as follows: Where, ∆rGo = cha...
Q: alculate pressure of gas collected over water Magnesium metal is reacted with hydrochloric acid to p...
A: Given - - > Hydrogen gas which is collected over water in eudiometer = 28°C Atmospheric pressure ...
Q: Sevee 1 Conas hd e MISSED THIS? Watch MEA4 Read Section 3 You can cick on the Review ink to access t...
A:
Q: estion 48 What is the molar solubility of Agl Ksp 8.3 × 101 in the presence of 0.05 M KI? O 9.11 x 1...
A:
Q: A mixture of NaOH, NaCl, and MgCl2 contains 34.6% by mass chloride and 20.0% by mass hydroxide. What...
A:
Q: In the year 1980, countries near the United States experienced an increase in acid precipi- tation. ...
A: The burning of Fossil fuels produces acidic oxide like sulfur dioxide or nitrogen dioxide etc. which...
Q: ACCURATE ERRONEOUS IMPOSSIBLE 2,2-dibromo-2,3- nonanediol ethene
A: For the IUPAC name, rules are there and when these rules are not followed, it will be wrong naming.
Q: How many milliliters of 0.136 M NAC2H3O2 are needed to supply 11.9 g NaC2H3O2? i mL
A:
Q: QUESTION 8 Calculate the pH during the titration of 25.00 mL of 0.1000 M LiOH(aq) with 0.1000 M HI(a...
A: Concentration of LiOH = 0.1000M Volume of LiOH=25.0mL Concentration of HI = 0.1000M Volume of HI = 2...
Q: step-s H3 O+ or anya Na oci codium hypochlocite acia protanated hypochlogous a cid step-2 (acia) HE ...
A: Answer: In this reaction we are preparing cyclohexanone by the hypochlorite oxidation of cyclohexano...
Q: Calculate the theoretical percentage of water in Cu2SO4·2H2O (Molar mass of the hydrate is 259.19 g/...
A:
Q: Which of the following bases would you not use to deprotonate 1- pentyne? B H3C H3C CH2 lithium n-bu...
A: Terminal Alkyne has acidic hydrogen which can be removed by using strong base.
Q: Calculate the pH for a 284 x 10 M HCIO solution. Consider the activity in your calculation. 1. What ...
A:
Q: A container of O2 gas at 75 °C has a volume of 3.5 liters at 1.3 atm. What is the new volume of the ...
A: Given :- Initial pressure (P1) = 1.3 atm Initial volume (V1) = 3.5 L Final pressure (P2) = 0.80 at...
Q: n which solution PbSO4, be most soluble ? Why? 1 pure H2O 2. 0.05mol/L NaNO3 3. 0.05mol/LNa2SO4 ...
A: A multiple choice question based on solution that is to be accomplished.
Q: 4. What is the temperature change for 500 g of each meta below, when 1,000 j of energy is added to e...
A: Few questions based on thermochemistry that is to be accomplished.
Q: If 500mL of 0.1IF lead altrate solution is added to 1L of 0.25F potossium lodate, calculate : a. num...
A:
Q: Ethylenediaminetetraacetate(EDTA*) is used as a complexing agent. Solutions of EDTA¯* are used to tr...
A:
Q: Combining 0.315 mol Fe,0, with excess carbon produced 18.0 g Fe. Fe,0, + 3C - 2 Fe + 3 CO What is th...
A:
Q: Which product would be obtained if 3-methyl-2-butanol was used as the alcohol in the Electrophilic A...
A:
Q: Experiment 3 Repeat the experiment above (Question 2) using a 10 mL pipette to dispense the 10 mL of...
A: The mean weight, standard deviation and the RSD of the water dispensed is needed to be calculated. ...
Q: Identify the equilibrium-constant expressions for the following reactions 2H2S(g) -> 2H2 (g) + S2(g)
A: For gaseous state, Equilibrium constant i.e. Kp = [pProducts][pReactants]
Q: Given: 10 g of o-hydroxybenzoic acid (density: 1.44 g/mL, MW: 138.2) 20 mL wood alcohol (density = ...
A:
Q: Solve all parts otherwise I will downvote...
A: A student has 5.35 g of zinc powder, 1.05 L of a 1.90 M calcium nitrate solution, and 1.70 L of a 2....
Q: Calculate the concentration of sulfide ions in a solution that is 0.050M in hydrogen sulfide and con...
A:
Q: Calculate the pH when 1.03 g of NaClO (FW = 74.44 g/mol) is added to 44.4 mL of 0.50 M HClO. Ignore ...
A: Answer: In this question we have to calculate the pH of the buffer made of HClO and NaClO.
Q: Na SOH SO-H NANO2 2-паphthol MW 144.17 HCI NH2 NO II NaOH p-sulfanilic acid MW 173.19 a diazonium sa...
A:
Q: On the basis of Kc for the following reaction, would you expect the reaction to go almost completely...
A: N2(g) + 2O2(g) <----> 2NO(g) Kc = 3 × 10-17 On the basis of Kc , would reaction go almost c...
Q: James is climbing a mountain. Is the distance he travels to the top of the mountain a state function...
A: A question based chemical thermodynamics that is to be accomplished.
Q: If OH- is added to a solution containing 1.2 g of Mg2+ and 1.28 g of Cu*2 per liter, which ion will ...
A:
Q: Direction: Answer the following. Write your solution and box the final answer. Topic: thermochemistr...
A:
Q: Q29) Match the following chemical terms to the descriptions: C Effervescent I Aliquot O Catalyst D E...
A: 1. small portion (of a reaction) - mother liquor 2. The substance dissolved in a solvent- solute ...
Q: High-purity benzoic acid (CH;COOH; AHxn for combustion =-3227 kJ/mol) is used as a standard for cali...
A: The heat of combustion of benzoic acid is = -3227 kJ/mol The mass of benzoic acid is = 1.221 g The h...
Q: Why use plastic reagent bottle for the storage of the standard NaOH solution instead of typical glas...
A: NaOH is a base. It is hygroscopic in nature. Hygroscopic means tends to absorb moisture from air. I...
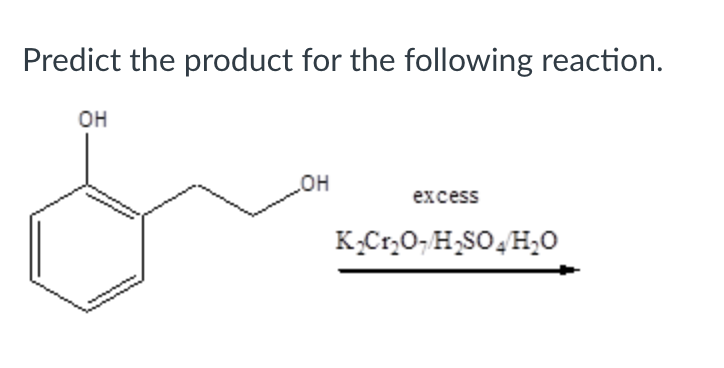
Step by step
Solved in 2 steps with 1 images


