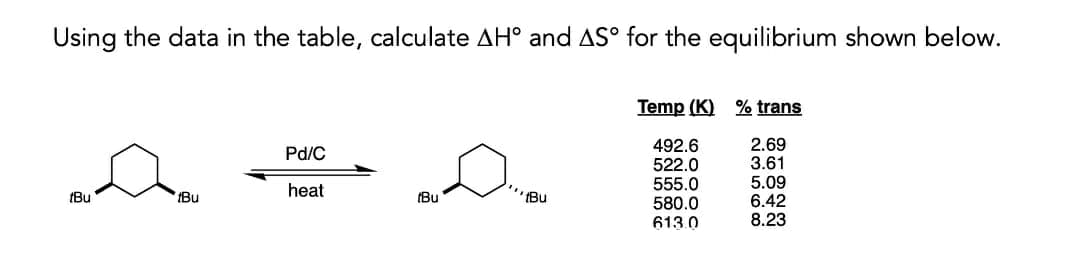
Using the data in the table, calculate AH° and AS° for the equilibrium shown below. Temp (K) % trans 492.6 522.0 555.0 580.0 613.0 2.69 3.61 5.09 6.42 8.23 Pd/C heat (Bu Bu (Bu 'Bu
Q: ΔG
A: Dear student I have given answer to your question in the image format.
Q: Given the following data, calculate ΔrH∘ for the reaction: N2O(g)+NO2(g)⟶3NO(g)…
A: The enthalpy change is a state function and the Hess law states that the change of enthalpy in a…
Q: Calculate AHo for the reaction: 20;Hlg) + 70x(g) - 4 CO:g) + 6 HO(g) If. CHi(g) + 30:(9) -2 CO:(g) +…
A:
Q: a) Calculate the AH far the fellowiveg reaction usivng Heas' Law: i) CS 2+2H2O +COz+ 2H2S Gilen…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Given the following AH° 298 K/(kJ/mol) values, 4 NH3 (g) + 5 02 (g) → 4 NO(g) + 6 H2O(I) 2 NO(g) +…
A: This question can be solved using Jess's law of constant heat summation.
Q: 7. Calculate the AHReaction for the reaction 2H2(g) + CO(g) CH3OH(/) Using the following equations:…
A: Given that - CH3OH(l) + 1/2O2(g) ----> C(s) + 2H2O(l) , ∆H1 = -304.97 kJ ----(1) C(s) +…
Q: 1. You have calculated the AH for the formation of ammonium chloride from your experimental data HCI…
A: Given Reaction : HCl + NH3 ---> NH4Cl ∆H°f = -167.159 KJ/mole ∆H°f = -80.835 KJ/mole ∆H°f =…
Q: How do I solve this
A:
Q: Given the following data: Pals) + 6 Clzig) → 4PC|3(9) AH = -1225.6 kJ Pa(s) + 5 Oz(g) → P4O10/s) AH…
A: enthalpy is an extensive property which will depend on the number of particle.
Q: . Does your answer agree with what you would predict based on bond energies? Explain.
A: The nature ie endothermic or exothermic of a chemical can be determined by calculate bond energy of…
Q: In predicting which side of an equilibrium is more favored, the factor concerned with probability is…
A: A question based on equilibrium concept that is to be accomplished.
Q: Given the following information: 2 A(g) + B(g) ⇌ A2B(g) K1 = 5.0 x 104 2 A(g) + C2(g) ⇌ 2 AC(g) K2 =…
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: Kc for 2N20 (g) = 2N2 (g) + O2 (g) is 7.3 × 1034. What is K for the following reaction, at the same…
A: Equilibrium constant is the ratio of product of concentration of products to that of the reactants…
Q: Which of the following is the most stable towards heat? * O plastic O sugar O benzoic acid O sodium…
A: Heat stability is the property of a molecule to retain it's structure intact in high temperature.
Q: Calculate the standard enthalpy for the reaction: 4NH:(g) + 5O:(g) → 4NO(g) + 6H;O(g), through:…
A:
Q: Calculate AH for the reaction C2H4 (g) + H2 (g) → C2H6 (g), from the following data. C2H4 (g) + 3 O2…
A:
Q: 8. Given that S(g) + O2(g) →→ SO2(g) 2 S(g) + 3 O2(g)→ 2 SO3(g) calculate AG° of the following…
A: Among the given reactions, multiplying reaction 2 with 0.5 and than subtracting reaction 1 from it…
Q: Q2A. Calculate the Equilibrium constant (Keg) for the following reaction:
A:
Q: The values of AH° (298 K) for SO2 and SO3 are -296.8 and -395.7 kJ/mol respectively. What is the…
A: The enthalpy change of a chemical reaction can be calculated by the standard heat of formation of…
Q: The reaction 4Al (s) + 302 (g) → 2 Al,O3 (s) AH° = -3351 kJ %3D is and therefore heat is by the…
A: The reaction in which heat release in the form of energy into the environment, is called an…
Q: Photosynthesis in plants converts carbon dioxide and water into glucose (C,H1206) and oxygen…
A: Dear student, Since you have posted multiple questions, the answer for first question is given…
Q: Calculate the AGᵒrxn using the following information at 25 °C 7 N₂(g) + H₂O(1) HNO3(g) + N₂H4(1)…
A:
Q: eaction: ZnO (s) + C (graphite) = Zn (s) + CO (g) Heat of fusion of zinc at melting point (420°C) =…
A: Enthalpy is the heat content of a system. Entropy is the measure of a systems thermal energy…
Q: H2 (9) + 2 02 (9) 4 H2O (1) 3. Calculate AH for the reaction CH4 (g) + NH3 (g) HCN (g) +3 H2 (g),…
A: Given:
Q: 6. When heat is added to a system at equilibrium and the reaction shifts to the left, which…
A:
Q: Consider the following reaction at 25 °C: 5 SO:(g) + 2 NH:(g) → 2 NO(g) + 5 SO:(g) + 3 H20(g) Given…
A:
Q: Calculate the value of ?p for the equation C(s)+CO2(g)↽−−⇀2CO(g)?p=? given that at a…
A: The equilibrium constant for reaction Kp1 = 3.37 The equilibrium constant for reaction Kp2 = 0.657…
Q: Determine AG° for the reaction 3 N,(g) + 2 O3(g) 6 NO(g) at 223°C. AH° = +257.7 kJ/mol kJ/mol AS° =…
A: ∆G° for the below reaction at 223°C needs to be determined 3 N₂(g) + 2 O₃(g) →…
Q: 5. The AH⁰ for the reaction (Na2O(s) + SO3(g) → Na2SO4(g)) equal to: Na(s) + H₂O() → NaOH(s) + 1/2…
A: As you have asked multiple question. But we will answer all the parts.
Q: Calculate ASO for the reaction SICI4(g) + 2Mg(s) → 2MgCl2(s) + Si(s) Substance SICI4(g) Mg(s)…
A: Entropy (S) :- Entropy is a measure of disorderness or randomness in a system. The change in…
Q: 6. Calculate the formation enthalpy (4H orm) for benzene (CSH6). Is this reaction endothermic or…
A:
Q: What is the S for the reaction? SO3(g) + H2O(1) –> H2SO4(1) 69.9 Given: S°(J/K mol): 256.2 156.9 O +…
A: The balance chemical reaction : SO3(g) + H2O(l) ---> H2SO4(l) S°(J/k.mol):…
Q: What is DH°rxn (in kJ) for the reaction - CH4(g) + 202(g) · CO2(g) + 2H20(1). --> DH°f (CH4(g))…
A:
Q: From the data below calculate G for the next reaction (at 298 K) 2CO (g) + O2(g) --> 2CO2(g)
A: The relationship between ΔG for a reaction and ΔH and ΔS is as shown below. ΔG = ΔH - TΔS where ΔG =…
Q: (a) 2 FeCl3(s) + 3 H2O(g) (equilibrium arrows) Fe2O3(s) + 6 HCl(g) Keq = (b) 3 NO(g)…
A:
Q: 12. For the reaction Cl,0(g)+%O2(g) → 2C10, (g), AH° = 126.4 kJ/mol and AS° =-74.9 J/K mol. At…
A: Given Data:
Q: Estimate AH for the reaction. нн нн H-S-C-C-S-H+2H–Br Br—C—С—Br + 2 H—S—Н нн нн
A: Given average bond enthalpies : Bond∆Hbond(KJ/mol)C-H413 C-S259S-H339H-Br366C-Br276C-C…
Q: Calculate AG° for the following reaction using the table below. Do not include units with your…
A: The ∆GRxn0 will be calculated using the following relation.…
Q: A 641.003 g block of Cu at 661.659 °C is plunged into 1.204 kg of water (T = 58.134 °C) in an…
A: Given: Mass of water = 1.204 kg = 1204 g Initial temperature of water (Ti) = 58.134 ℃ Mass of Cu…
Q: Oxygen was first prepared by heating mercury(II) oxide, HgO. 2H9O(s) 2Hg(9) + O2 (9) Estimate the…
A: The balanced chemical equation can be expressed as: The decomposition of mercury oxide has to be…
Q: Calculate the AS for the given reaction. → Ca3(PO4)2(») 2+ Ca +PO,3- (ag) (ag) molar ratio of the…
A:
Q: Given the following AH values, calculate AH for the reaction 6 CO, + 6 H,0 → C,H12O6 + 6 Oz AH;…
A: The reaction which requires energy from outside is called an endothermic reaction while the reaction…
Q: QUESTION 6 Özone (O 3) in the atmosphere can react with nitric oxide (NO): O 3(g) + NO(g) NO 2(g) +…
A: 6. Given information, Temperature is 250C = 298 K∆H0 = - 199 kJ/mol and ∆S0 = - 4.1 J/K…
Q: Given the following equation, C3HS(g) + 5 O2(8)→3 CO2(g) + 4 H2O(g) AG pan-2074 k] Calculate AGpn…
A: ∆Grxno for a chemical reaction have same value with it's reverse reaction but having opposite sign…
Q: Determine AG for the reaction: 2C0(g) + 2H2(g) → CO2(g) + CH4(9) AH =-247.3 kJ/mol AS -256.5 J/mol K…
A:
Q: Given the following data 2 CIF(9) + O2 (g) Cl2 O(g)+ F20(g) 2 CIF3 (g) + 202(9) → Cl2 O(g) + 3F,0(g)…
A:
Q: CO₂Et ANO2 Pd catalyst Et N, heat
A: Note: According to our guidelines we are supposed to answer only first question. Kindly repost other…
Q: What is vahse or equilliorum constant for Pollowns? (AP)+ HF(AP) + Yencut) or HE) Kb
A: Equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical…
Q: Expain the dynamic equilibrium.
A: Equilibrium can be classified as physical equilibrium and chemical equilibrium. The equilibrium set…
Q: Is the following chemical reaction exothermic or endothermic? 31 (aq) + H3AS04(aq) + 2H* (aq) 13…
A: Answer: The chemical reaction is endothermic.
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 1 images

- If Sº in J/mol-K is 20 for H2SO4(aq), 20 for SO42-(aq), 141 for NaBr(aq), & 59 for Na+(aq), Sº for HBr(aq) must be about a. undetermined since it must be measured empirically b. 102 J/mol-K c. 79 J/mol-K d. 82 J/mol-KCompute for the PCl5 concentration in the following reaction at 78°C equilibrium. PCl3(g) + Cl2(g) ↔ PCl5(g) Kc = 3.572 x 104 PCl3 = 0.238 Cl2 = 0.168Consider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 1.87 × 10⁻³ at a certain temperature. If 5.00 g of solid PH₃BCl₃ and 0.0700 g of BCl₃ are added to a 4.500 L reaction vessel, what will the equilibrium concentration of PH₃ be? PH₃BCl₃(s) ⇌ PH₃(g) + BCl₃(g) 1.) Set up the expression for Qc and then evaluate it to determine the direction of the reaction. Do not combine or simplify terms 2.) Based on the given values and your value for Qc, set up ICE table in order to determine the unknown. 3.) Based on your ICE table, set up the expression foe Kc in order to determine the unknown. 4.) Based on your ICE table and expression for Kc, solve for the partial pressure of PH3, at equilibrium.
- Based on a Kc value of 0.170 and the data table given, what are the equilibrium conversations of XY, X, and Y, respectively?Determine the equilibrium constant (Kc): data is attached if neededConsider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 4.10 × 10⁻⁴ at a high temperature. If 0.20 mol N₂ and 0.15 mol O₂ react in a 1.0 L vessel, what will the equilibrium concentration of O₂ be? N₂(g) + O₂(g) ⇌ 2 NO(g) A )Set up the expression for Qc and then evaluate it to determine the direction of the reaction. Do not combine or simplify terms. Qc = _____/_____ = _____ Based on the given values and your value for Qc, set up ICE table in order to determine the unknown. Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms. Kc = (________) / (_______)= 4.10 × 10⁻⁴ Based on your ICE table and expression for Kc, solve for the concentration of O₂ at equilibrium. [O₂]eq = _____M
- Consider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 4.10 × 10⁻⁴ at a high temperature. If 0.20 mol N₂ and 0.15 mol O₂ react in a 1.0 L vessel, what will the equilibrium concentration of O₂ be? N₂(g) + O₂(g) ⇌ 2 NO(g) Set up the expression for Qc and then evaluate it to determine the direction of the reaction. Do not combine or simplify terms. Qc= (_______) / (_______) = (______) Based on the given values and your value for Qc, set up ICE table in order to determine the unknown. N₂(g) + O₂(g) ⇌ 2 NO(g) Initial (M) Change (M) Equilibrium (M) Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms. Kc = (________) / (_______)= 4.10 × 10⁻⁴ Based on your ICE table and expression for Kc, solve for the…What would be the equilibrium temperature if you drop 5.0 g of brass (Cp= 0.38 J/g-°C, initially at 68°C) and 3.5 g of aluminum (Cp = 0.9 J/g-°Cinitially at 83°C) into 105 g of water (Cp = 4.184 J/g-°C,initially at 23°C)?Dinitrogen tetroxide is 18.46 per cent dissociated at 25°C and 1.00 bar in the equilibrium N2O4(g) ⇌ 2NO2(g). Calculate K at (i) 25°C, (ii) 100°C given that ΔrH° = +56.2 kJ mol−1 over the temperature range.
- Compute for the PCl5 concentration in the given reaction at 78°C equilibrium. PCl3(g) + Cl2(g) ↔ PCl5(g) Kc = 3.572 x 104 PCl3 = 0.239 Cl2 = 0.169What would be the equilibrium temperature (oC) if you drop 5.0 g of brass (Cp = 0.38 J/g-oC, initially at 68oC) and 3.5 g of aluminum (Cp = 0.9 J/g-oC, initially at 83oC) into 20 g of water (Cp = 4.184 J/g-oC, initially at 23oC)?A 348.0 g block of Cu at 639.0°C is plunged into 1.47 kg of water (T = 27.4 °C) in an insulatedcontainer. What will be the final equilibrium T of the water and the Cu? (cCu = 0.385 J g-1°C1, cH2O = 4.184 J g-1°C-1)



