Q: Rank the following in order of increasing energy. Lowest energy first. O B<A<C OA<C<B OA<B<C O B-C<A...
A: This can be explain by resonance effect, higher the resonance less will be the energy. here C will b...
Q: 4.32 (a) Calculate the enthalpy of formation of the hypothetical compound KF, assuming a CaF, struct...
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A:
Q: The Ka for HF is 7.0 x 10-4. Kh for the fluoride ion is O 1.6 x 103 O 1.4 x 10-11 O 7.0 x 10-18 O 2....
A: Ka is the acid dissociation constant and Kb is the base dissociation constant. Higher value of Ka i...
Q: 5.00 mol of a perfect gas expand isothermally at 298 K from 2.00 dm3 to 12.00 dm3 Calculate the wor...
A:
Q: Identify the Brønsted-Lowry acid, the Brønsted-Lowry base, the conjugate acid, and the conjugate bas...
A: Identify the bronsted lowry acid, base, conjugate acid and conjugate base in the following reaction ...
Q: Disulfide dichloride (S,Cl,) is used in the vulcanization of rubber, a process that prevents this sl...
A:
Q: OH он toluene H OH 130 °C OH HO, ÓH Ph "I |
A:
Q: A solution (50.00 mL) of citric acid (a weak acid with concentration 0.0950 M and Ka = 1.8 × 10−5) w...
A:
Q: 10. What is the molarity of 750 mL of a solution that contains 40.0 g of dissolved CUSO,
A:
Q: A+B+C → D Reaction [A] (М) [B] (М) [C] (М) Experiment M. Rate (-) 1 0.10 0.10 0.10 0.003 2 0.20 0.10...
A: The given reaction is as follows: A + B + C → D Let the order of the reaction with respect to A, B, ...
Q: Decide whether the following molecule presents an S or R configuration for the stereogenic carbon(s)
A: RS nomenclature: The RS nomenclature is an important nomenclature system for denoting the configurat...
Q: : Identify all the symmetry elements in the following solid-state structures: a. a body-centered tet...
A: The symmetry elements present in the given solid state structures. A symmetry elements is a point, ...
Q: If you have 72 g of sodium sulfate, calculate the sodium, sulfur, and oxygen atoms that are present ...
A: Formula of sodium sulfate - Na2SO4 Molar mass of sodium sulfate - 142.04 g/mol Avogadro's constant -...
Q: Suppose that 2.5 ml of 20% AgNO3 solution (density is 1.19 g/ml) was used to precipitate the chlorid...
A: Given, Mass percentage of AgNO3 in solution = 20% Volume of AgNO3 = 2.5 mL Density of the solutio...
Q: 5.3 QUESTION 7 Fill in the table (round mass and all answers to 3 sig fig) Mass Number of atoms 1 at...
A:
Q: NH2 major product?
A:
Q: Which is longer, the carbon-oxygen single bond in a carboxylic acid or the carbon-oxygen bond in an ...
A:
Q: Write the balanced NET ionic equation for the reaction when cesium hydroxide and sulfurix acid are m...
A: The net ionic equation can be defined as the chemical equation which involves only the actual ions o...
Q: On the mountain in Kalinga ,a mountain biker carried an aneroid barometer in his bike from base to t...
A: Given: Barometric pressure on the ground = 35.12 in.Hg Barometric pressure on the mountain = 26.807 ...
Q: What is the pH of a 0.250 M ammonia solution? Express your answer numerically to two decimal places....
A:
Q: How would you carry out the following reactions
A: Given reaction is aldol condensation followed by oxidation
Q: 1. Illustrate the mechanism for the following reaction. OH H2 SOy 200 °C
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please re...
Q: An organic compound was prepared and purified by chromatography. A conductivity measurement showed t...
A: Temperature = 20℃ = 20 + 273 = 293 K The molar concentration of the solution is calculated: Using Id...
Q: Give two possible [1,2]-rearrangement products where an alkyl group migrates in the following compou...
A: Organic reaction mechanisms:
Q: Buli SnMeg
A: EXPLANATION : Given reaction is 2,3-sigmatropic rearrangements reaction. 2,3-sigmatropic rearra...
Q: Is the use of an indicator to determine a concentration using titration as good as using a pH meter?...
A: The explanation of the following is:
Q: What is the least volume of 3.10 M HCl(aq) that will exceed the buffer capacity of 1.00 L of a buffe...
A:
Q: Identify the method of sterilization/ chemical agent described A method of sterilization that invol...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: Match the correct response When the pH of a solution rises what happens to the followings [H3O+] A. ...
A:
Q: Rank the solutions in order of decreasing H3O|.
A: Acids donate [H+] ions and increase H+ ions concentration in the solution Strong acids dissociat...
Q: Calculate the entropy change of the surroundings when 1 mol of a perfect gas expands isothermally (a...
A:
Q: The following equation shows the equilibrium in an aqueous solution of methylamine: CH3NH2 (aq) + H2...
A: Given : CH3NH2(aq) + H2O(l) <----> CH3NH3+(aq) + OH-(aq) To identify :- Conjugate acid-base...
Q: A 13.3 L scuba tank contains a mixture of nitrogen, oxygen, and argon. 72% is nitrogen, 4% is argon,...
A: Answer: In this question, composition of each gas in scuba tank is given and we have to find out exa...
Q: OH 2 cat. Me -0 H;C НО H3C CH3
A: Requirement from question Draw given reaction using any tool:
Q: 1-Bromopropane is synthesized by treating 1-propanol with sulfuric acid (H2SO4) and potassium bromid...
A:
Q: How would you carry out the following reactions
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: Aqueous sulfuric acid (H,So,) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium su...
A: Answer: 67.0 %
Q: [OH-] = 2.3 × 10-10 Express the concentration to two significant figures and include the appropriate...
A:
Q: Rank the following compounds in order of decreasing acid strength using periodic trends. Rank the ac...
A:
Q: Common aspirin is acetylsalicylic acid, which has the structure shown below and a pKapKa of 3.5. Ca...
A: Given data : Mass of Aspirin = 670 mg water = 10 ounces pKa of Aspirin = 3.5 Molar mass of Aspirin =...
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NAOH) to produce aqueous sodium b...
A:
Q: An impure sample of calcium carbonate with a mass of 7.95 g was reacted with 50.00 cm3 of 1.00 mol d...
A: Given: mass of sample = 7.95 g Concentration of HCl = 1.00 mol.dm-3 Volume of HCl = 50 cm-3 = 0.05 d...
Q: DIBALH, THE,-78°C - major product? O Hzo
A:
Q: CO,Me OSIME, CO,Me Br
A: We have to draw the mechanism. And also we have to tell about the stereochemist of the compound and ...
Q: PRE-LAB QUESTION After consulting sections 5.7 of your text, draw the complete MO diagram for the di...
A:
Q: Reaction Mechanism for the Synthesis of 1,1’-(buta-1,3-diyne-1,4-diyl)di(cyclohexan-1-ol) from 1-eth...
A: It is coupling reaction between terminal alkyne. It is carried out in presence of CuCl and TMEDA as ...
Q: Draw the major product from this reaction. Use wedge and dash bonds to indicate relative stereochemi...
A: We have to draw the major organic product of the given reaction.
Q: он OEt cinnamaldehyde cinnamic acid ethyl cinnamate 1. Dissolve in ether 2. Add aq. NaOH aqueous org...
A:
Q: The two chemical reactions that occur in this in the Percentage of Copper in Copper(II) Sulfate expe...
A: In the determination of Copper in Copper sulfate experiment we find the mass of CuSO4 obtained from ...
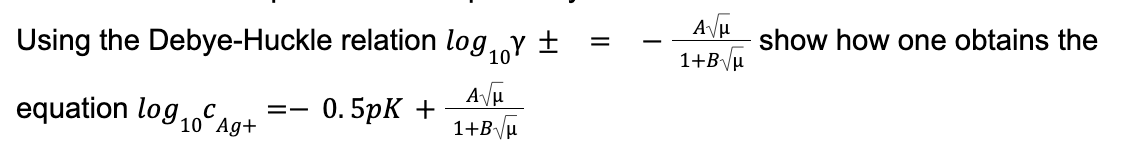
Step by step
Solved in 3 steps

- Describe the primary and secondary criteria for having achieved V ̇O2 max.Derive a general expression that shows pH dependence of the distributioncoefficient defined in (2-20) for a weak acid between a fermentation broth and an organic solvent.If an instrument gives a response of 1240 for a standard containing 8 ppm of a substance, how much if this substance is in a sample that gives a response of 1705? Are any assumptions needed?
- A solution of 710.5 ml of 0.00056 M A3B2 is mixed with a solution of 502.5 ml of 0.00044 M C1D2. Find the value of pQ (the minus log of Q) for the formation of C3B2.How is the operation of an ion selective probe used to measure pH. How does it specifically relate to the probe interface?A 0.1475-M solution of Ba(OH)2 was used to titrate the acetic acid (60.05 g/mol) in a dilute aqueous solution. The following results were obtained. (See attached image)(a) Calculate the mean w/v percentage of acetic acid in the sample.(b) Calculate the standard deviation for the results.(c) Calculate the 90% confidence interval for the mean.(d) (d) At the 90% confidence level, could any of the results be discarded?
- y= mx+ b Slope () = -4E-09 y-intercept () = 0.4021 and OD= 0.540 , Calculate the concentration (x)Which of the following would result in an increase in the calculated ngas 10, assuming the rest of the parameters are held constant? I. Inserting 23.0 mg instead of the prescribed 15.0mg of folded Mg ribbon into the eudiometer. II. Performing the experiment in a different laboratory with a cooler temperature. III. A shorter equilibration time prior to volume measurement. IV. Acid used was 12.0 M HNO3 instead of 6.0 M HCI.If 5 µM enzyme was used to obtain the data in the summary plot below, V-max = ________________ µM sec-1 and Km = ____________ µM for this enzyme (Enter numeric values to the nearest integer; Do NOT write units.)
- Identify the effect of the condition on the indicated parameter. Write increase, decrease, or no effect , and provide an explanation The actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value. However, the NaOH solution used in the determination of ΔHrxn was of the correct concentration. [magnitude of ΔHrxn]From the equivalent conductivities at infinite dilution following at 18°C, find the value of Λ° for NH4OH:Ba(OH)2 Λ° = 228.8BaCl2 Λ° = 120.3NH4Cl Λ° = 129.8Which of the following would result in an increase in the calculated ngas in the Chemistry Experiment, assuming the rest of the parameters are held constant? I. Inserting 23.0 mg instead of the prescribed 15.0mg of folded Mg ribbon into the eudiometer. II. Performing the experiment in a different laboratory with a cooler temperature. III. A shorter equilibration time prior to volume measurement. IV. Acid used was 12.0 M HNO3 instead of 6.0 M HCl. a. II and III b. I and II c. I only d. II, III, and IV



