Using the equations N2 (g) + O2 (g)→ 2 NO (g) AH° = 180.6 kJ/mol N2 (g) + 3 H2 (g) → 2 NH3 (g) AH° = -91.8 kJ/mol 2 H2 (g) + O2 (g) 2 H20 (g) AH° = -483.7 kJ/mol %3D Determine the enthalpy for the reaction 4 NH3 (g) + 5 O2 (g)→ 4 NO (g) + 6 H2O (g).
Using the equations N2 (g) + O2 (g)→ 2 NO (g) AH° = 180.6 kJ/mol N2 (g) + 3 H2 (g) → 2 NH3 (g) AH° = -91.8 kJ/mol 2 H2 (g) + O2 (g) 2 H20 (g) AH° = -483.7 kJ/mol %3D Determine the enthalpy for the reaction 4 NH3 (g) + 5 O2 (g)→ 4 NO (g) + 6 H2O (g).
Chapter6: The States Of Matter
Section: Chapter Questions
Problem 6.9E
Related questions
Question
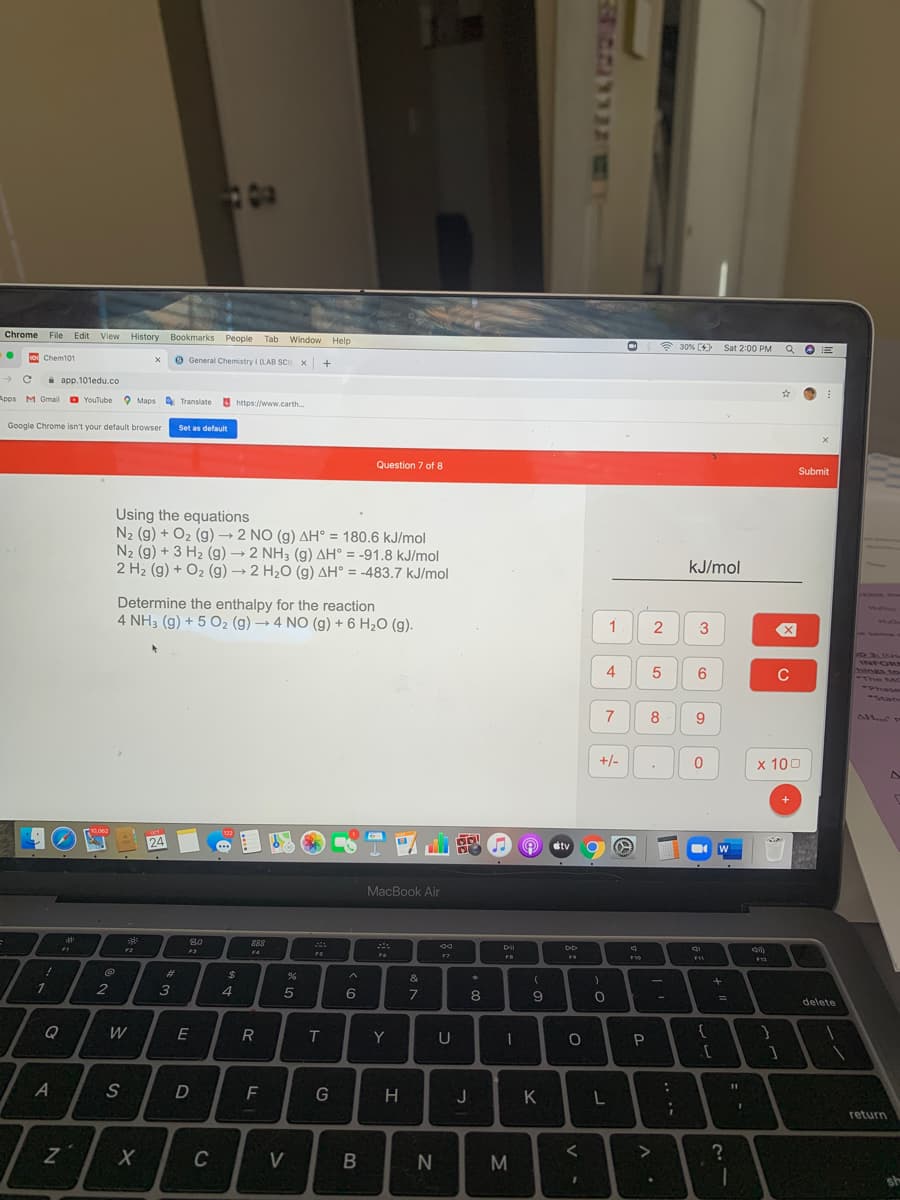
Transcribed Image Text:Chrome File Edit View History Bookmarks
People
Tab Window Help
* 30% (4) Sat 2:00 PM Q O E
0 Chem101
O General Chemistry I (LAB SC) x
i app.101edu.co
Apps M Gmail O YouTube Maps Translate
3 https://www.carth..
Google Chrome isn't your default browser
Set as default
Question 7 of 8
Submit
Using the equations
N2 (g) + O2 (g) → 2 NO (g) AH° = 180.6 kJ/mol
N2 (g) + 3 H2 (g) → 2 NH3 (g) AH° = -91.8 kJ/mol
2 H2 (g) + O2 (g) → 2 H2O (g) AH° = -483.7 kJ/mol
kJ/mol
Determine the enthalpy for the reaction
4 NH3 (g) + 5 O2 (g) → 4 NO (g) + 6 H2O (g).
1.
3
INFOR
4
6
Coings t
C
The MC
Uhase
Stan
8
9
+/-
x 100
+
10.062
122
24
tv
MacBook Air
888
DII
F4
F5
F7
F9
12
%23
24
&
*
2
4
6
8
%3D
delete
Q
W
E
R
Y
A
S
D
F
G
H
J
L
return
>
C
V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



