Using the following Latimar diagram, calculate the value that would be plotted on the Frost diagram for the NH4+ species. Have your answer to 2 decimal places.
Using the following Latimar diagram, calculate the value that would be plotted on the Frost diagram for the NH4+ species. Have your answer to 2 decimal places.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter13: The Chemistry Of Solutes And Solutions
Section13.7: Colligative Properties Of Solutions
Problem 13.16E: Suppose that you are closing a cabin in the north woods for the winter and you do not want the water...
Related questions
Question
Using the following Latimar diagram, calculate the value that would be plotted on the Frost diagram for the NH4+ species.
Have your answer to 2 decimal places.
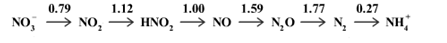
Transcribed Image Text:0.79
1.12
1.00
1.59
1.77
0.27
NO, → NO, –→ HNO, → NO → N,0 → N, → NH
Expert Solution
Step 1

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning