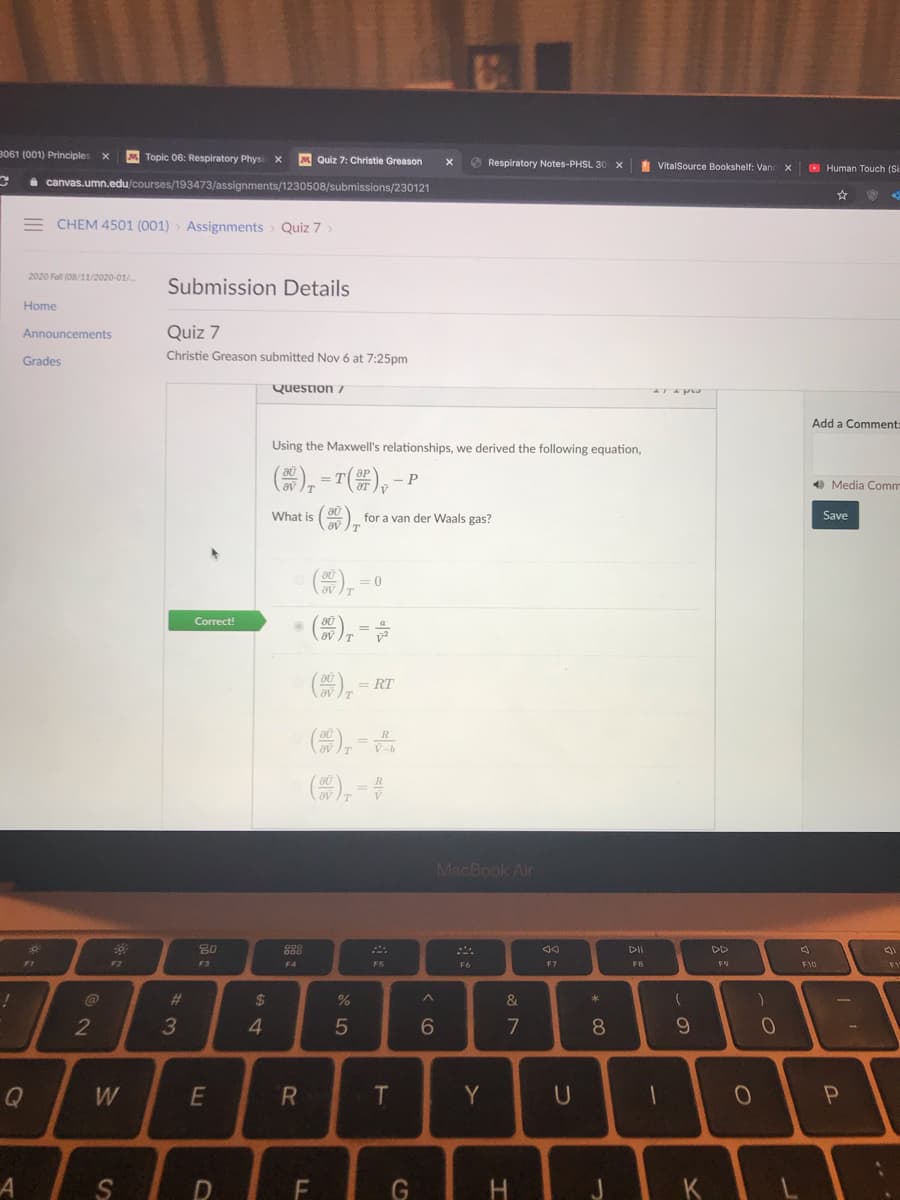
Using the Maxwell's relationships, we derived the following equation, ), ="(#),-P What is () for a van der Waals gas? = 0 (器),= = RT av (), - R Ov IT
Q: 25.0 mL Sampre of neon gas at AD 455 mm.Ha is culed Prom roo °C to 10C If the volume remaiins…
A: The pressure of the gas increases with increases in the temperature of the gas. Gay-Lussac's law…
Q: Calculate the average kinetic energy, in J/mol, for a mole of Xe at -23.0°C. Assume ideal gas…
A: The average kinetic energy of Xe at - 23°C is = 3117.75 J/mol
Q: Q4// A-Aclosed container of volume 0.02m contains a mixture of neon and argon at tempreture 27°c and…
A: Volume = 0.02 m3 Pressure = 1 × 105 Nm-2 Temperature = 27°C = 27 + 273 = 300 K Total mass of gases…
Q: What volume of carbon dioxide (in L) will 3.30 g of antacid made of calcium carbonate produce at…
A:
Q: one whic meteer volume of uranium heraf boride gas is cund, pressore of 1.5 atmospheres. t cohat is…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: In an experiment reported in the scientific literature, malecockroaches were made to run at…
A: The ideal gas expression is shown below: PV = nRT where, P = pressure of the gas V = volume of the…
Q: )The South le is aclually an elevation of 9000 ft, Go with a pressure ofE 10.5 psi,Ond the mole…
A: Given-> P = 10.5 psi Mole fraction = 0.00033
Q: Amixture of methane and hydrogen gases contains methane at a partial pressure of 505 mm Hg and…
A: Generally the mole fraction is determined by dividing partial pressure by total pressure. First…
Q: Calculate the mean speed of atoms of argon in a gas at 25oC if the mean speed of neon under these…
A:
Q: Assume that the atmosphere is at a constant temperature T and that the partial pressure of a gas…
A: On going at higher altitude air molecules become far apart to each each other due to which air…
Q: If both gases behave ideally, then what would be the average kinetic energy for 1.00 mol of either…
A: The question is based on the concept of kinetic theory of gases. we have to calculate average…
Q: The difference between C, and C, can be neglected in gases. True O False
A: The difference between Cp and Cv is R (universal gas constant).
Q: The ideal gas law is PV = NkT, with k denoting the Boltzmann constant. It is important that the…
A: Ideal gas law is obtained by combining all the Gas laws to derive a common relation among all the…
Q: Q4) How to obtain the ideal gas equation PV= nRT by Charles observation and by Boyles observation?
A: Boyle's law :- According to Boyle's law , at constant temperature the volume of certain mass of gas…
Q: The gas constant R has the value 287 J/Kg * K for dry air. Using the ideal gas law (P = PdryRT): a.)…
A:
Q: Q4// A-Aclosed container of volume 0.02m° contains a mixture of neon and argon at tempreture 27°c…
A: Given : Volume = 0.02 m3 Temperature = 27°C Pressure = 1×105 Nm-2 Total mass = 28 g To find : Masses…
Q: Place the following gases in order of increasing average molecular speed at 0.00 °C: Ar, NH3, CO,…
A: The formula for average molecular speed = uavg = 8RTπM The formula for root-mean-square speed =…
Q: A 1.52-gram sample of iron (III) perchlorate was decomposed according to the reaction below,…
A: First the moles of oxygen gas produced in the reaction is determined using ideal gas equation. Then…
Q: If it takes 6,45 seconds for S,4 mL Melg) gas to ef foze through a pin hole into o vacunm now many…
A: To calculate the time taken by 31.8 mL of H2 gas to escape through a pinhole.
Q: While resting, the average 70-kg human male consumes 14 L of pure O2 per hour at 25 °C and 100 kPa.…
A:
Q: Considering the ideal gas law PV = nRT what is P inversely proportional to ?
A: Given Ideal gas equation PV = nRT
Q: The Ideal Gas Law is given by the equation: PV = nRT Where: pressure V = volume n = moles T =…
A:
Q: A sample of hydrogen at 32°C exerts a pressure of 8.0atm. The gas is heat to 91°C, what will its new…
A:
Q: Explain how the function pv/RT can be used to show gases behave non-ideally at high pressure.
A:
Q: (b) Calculate the volume of oxygen at 39°C & 1200 torr pressure, when 170gm of KCIO: is heatec to…
A: The given balanced Chemical equation is - 2KClO3 -----> 2KCl + 3O2 Here two moles of oxygen gas…
Q: Which of the following statements about the Perfect Gas Law and the van der Waals equation of state…
A: Here, we have to find the statement that is false among the given statements according to the…
Q: To derive the ideal-gas equation, we assume that the volumeof the gas atoms/molecules can be…
A: To Find: To find the fraction of space that Neon atoms occupy in a sample of neon at STP.
Q: How would i calculate the tire pressure at 15.3 C uring the equation of the best fit line? What…
A: The data of pressure v/s temperature is given as,
Q: In a collision of sufficient force, automobile air bags respondby electrically triggering the…
A: The ideal gas law, also referred to as the general gas equation, is the equation of the state of the…
Q: Rank the following gases from least dense to most dense at1.00 atm and 298 K: CO, N2O, Cl2, HF.
A: The gaseous state is the state of matter with least intermolecular force of attraction between…
Q: At what temperature (in °C) would one liter of ethene gas, C2H4, have a mass of 1.303 g, if the gas…
A: Temperature at which ethene gas have mass 1.303 g and pressure 0.9382 atm when it behaves ideally…
Q: s volume if th
A:
Q: At what temperature (in °C) would one liter of ethene gas, C2H4, have a mass of 1.166 g, if the gas…
A: A mole of a substance refers to the mass of that substance which will contain same number of atoms…
Q: Consider the reaction: Li2O(aq) + CO2(g) Lİ2CO3(s) ----> What volume of cabon dioxide will react…
A: Given :- Li2O(aq) + CO2(g) ---> Li2CO3(s) Mass of Li2O = 1.00 g Molar mass of Li2O = 29.88…
Q: 4) Gliven the following seto of values for thee gas. variables, calaulate the unkaown quantty: La.…
A: In this question ( #55 ) we want to determine the mass of He and O2 gas. You can see details…
Q: the kinetic theory of gases is an attempt to explain the properties(behaviors} of ideal gases. what…
A: For the assumption of ideal gas behaviour representation , one theory arise , that theory is known…
Q: if a steam reforming line processes methane at a temperature fo 725.0 degrees and atmospheric…
A: According to ideal gas equation, PV = nRTwhere P is the pressure in atmospheres, V is the volume in…
Q: Patrences In 1897 the Swedish explorer Andree tried to reach the North Pole in a balloon. The…
A: Volume of balloon = 4800m3. % loss of Hydrogen = 20% Therefore, volume of Hydrogen to be released to…
Q: 1. Which one of the three varlables: Pressure, Volume or Temperature cannot be changed in Boyle's…
A: To answer questions regarding Boyle's Law.
Q: Qy - Boyle's Law says that the Volume V of a gas at Constant temperatune increases w henever the…
A: Given, P1 = 14.7 lbs/in2 , P2 = 23.4 lbs/in2 , V1 = 1000 in3 , V = ?
Q: At 350 °C and 101.325 kPa, the volume occupied by 48.00 mol of NH3(g) is a.bc × 10ª L. The values of…
A: From gas laws, we know: PV = nRT Where, P = pressure V = Volume N = number of moles of gas R = gas…
Q: What volume of carbon dioxide (in L) will 3.26 g of antacid made of calcium carbonate produce at…
A: CaCO3 (s) + 2HCl (aq) → CaCl2 (aq) + H2O (l) + CO2 (g) It is given that,mass of CaCO3 = 3.26 g…
Q: A storage tank of volume V contains the mass M of oxygen (O2) at pressure P. What is the temperature…
A: Given, Volume = V Mass of oxygen = M Pressure = P Required, Temperature…
Q: Rank the following gases from least dense to most dense at1.00 atm and 298 K: SO2, HBr, CO2.
A: Since we know that PM = dRT where P = pressure M = molar mass d = density R = universal gas…
Q: VOutme of day when the almosphienic pressure is arrives, and the atmospberic pressure drops to 0.962…
A: Given V1=8.5L P1=1.013bar P2=0.962bar Find the new volume that is V2 of the balloon. when the…
Q: reference (Ps 225 295) Section 82 while completing this problem art A J The volume of air in a…
A:
Q: 5. If you had1080 pressure, what would the volum e of the container be? gram s of a substance with…
A: 5. Given, Mass of a substance is ---1080g Its molecular mass is ---12.01g/mol Temperature is ---75oC…
Step by step
Solved in 2 steps

- 4. Ksp=4.1x10-36 for Pb3(AsO4)2Calculate Eofor Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO43-Pb2+ +2e- Pb(s) Eo= -0.126 VThe concentration of purified OXA-M290 is tested with a BCA assay. Serial dilutions of a bovine serum albumin (BSA) stock solution are prepared, then pipetted into a 96-well plate; each dilution of the BSA standard is tested in triplicate. Then, bicinchoninic acid and Cu2+ ions are added to all of the wells of the plate. After incubating the plate for 1 hour, a microplate reader is used to measure the absorbance of all of the wells in the plate at 560 nm. This generates the following data: BSA conc. (μg/mL), Replicate 1 Absorbance, Replicate 2 Absorbance, Replicate 3 Absorbance 40, 1.360, 1.403, 1.481 20, 0.750, 0.745, 0.810 10, 0.380, 0.344, 0.398 5, 0.198, 0.160, 0.183 2.5, 0.090, 0.100, 0.085 1.25, 0.038, 0.043, 0.051 0.625, 0.024, 0.028, 0.019 Prepare a calibration curve using these data. You can use Excel, R, SPSS or an equivalent graphing software. In this graph, plot absorbance (y-axis) against the concentration of the protein standard (x-axis). Calculate and plot…What is the simplest CGS unit for "poise"?
- You are trying to come up with a drug to inhibit the activity of an enzyme thought to have a role in liver disease. In the laboratory the enzyme was shown to have a Km of 1.0 x 10-6 M and Vmax of 0.1 micromoles/min.mg measured at room temperature. You developed a competitive inhibitor. In the presence of 5.0 x 10-5 M inhibitor, the apparent Km of the enzyme was found to be 1.5 x 10-5 M. What is the Ki of the inhibitor?Qq.1. Subject :- AccountConsider the following data. For the acid: H2A Ka = 4.21⋅10−44.21⋅10-4 Calculate the Kb for HA-. Report your answer in scientific notation using 3 sig figs.
- 10.5mL (mw 106) of benzaldehyde and 2.9g(3.63mL, mw 40) of acetone react with 5g of NaOH and 25mL of Ethanol to form 6.7g of Dibenzalacetone. What is the theoretical, actual and percent yield?There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…

