Using the working concentration of the standard glucose solution, the following standard solutions were prepared: Test Tube No. 1 2 W|N 3 4 45 5 Table 6.2. Preparation of the standard glucose solution Volume of 2.0 mM glucose solution (in mL) 0.00 0.15 0.30 0.50 0.70 Volume of dH₂O (in mL) 1.00 0.85 0.70 0.50 0.30 Q11. Determine the concentration of the prepared standard glucose solutions in test tubes 1-5. Tabulate your answers and express them in two decimal places.
Using the working concentration of the standard glucose solution, the following standard solutions were prepared: Test Tube No. 1 2 W|N 3 4 45 5 Table 6.2. Preparation of the standard glucose solution Volume of 2.0 mM glucose solution (in mL) 0.00 0.15 0.30 0.50 0.70 Volume of dH₂O (in mL) 1.00 0.85 0.70 0.50 0.30 Q11. Determine the concentration of the prepared standard glucose solutions in test tubes 1-5. Tabulate your answers and express them in two decimal places.
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
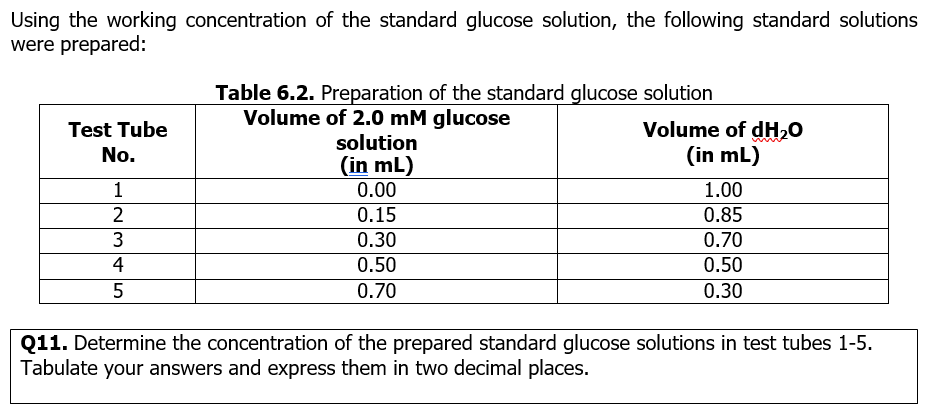
Transcribed Image Text:Using the working concentration of the standard glucose solution, the following standard solutions
were prepared:
Test Tube
No.
1
2
3
4
5
Table 6.2. Preparation of the standard glucose solution
Volume of 2.0 mM glucose
solution
(in mL)
0.00
0.15
0.30
0.50
0.70
Volume of dH₂O
(in mL)
1.00
0.85
0.70
0.50
0.30
Q11. Determine the concentration of the prepared standard glucose solutions in test tubes 1-5.
Tabulate your answers and express them in two decimal places.
![Nelson's test was first performed in standard glucose solutions to generate a linear equation to
determine the concentration of D-glucose hydrolyzed from the glycogen isolate. The linear equation is
constructed by plotting the concentration of standard glucose (in mM) on the x-axis against the
absorbance of the resulting mixture at 510 nm on the y-axis. As such, the general form of the linear
equation is:
Abs =
slope (in mM-1) [glucose] (in mM) + y - intercept](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Faab8bf0f-5ea1-445f-b855-c3e64d9a4705%2F362cab0a-3b6c-40f5-a725-e62f451d9201%2F6a5x4e8_processed.png&w=3840&q=75)
Transcribed Image Text:Nelson's test was first performed in standard glucose solutions to generate a linear equation to
determine the concentration of D-glucose hydrolyzed from the glycogen isolate. The linear equation is
constructed by plotting the concentration of standard glucose (in mM) on the x-axis against the
absorbance of the resulting mixture at 510 nm on the y-axis. As such, the general form of the linear
equation is:
Abs =
slope (in mM-1) [glucose] (in mM) + y - intercept
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

