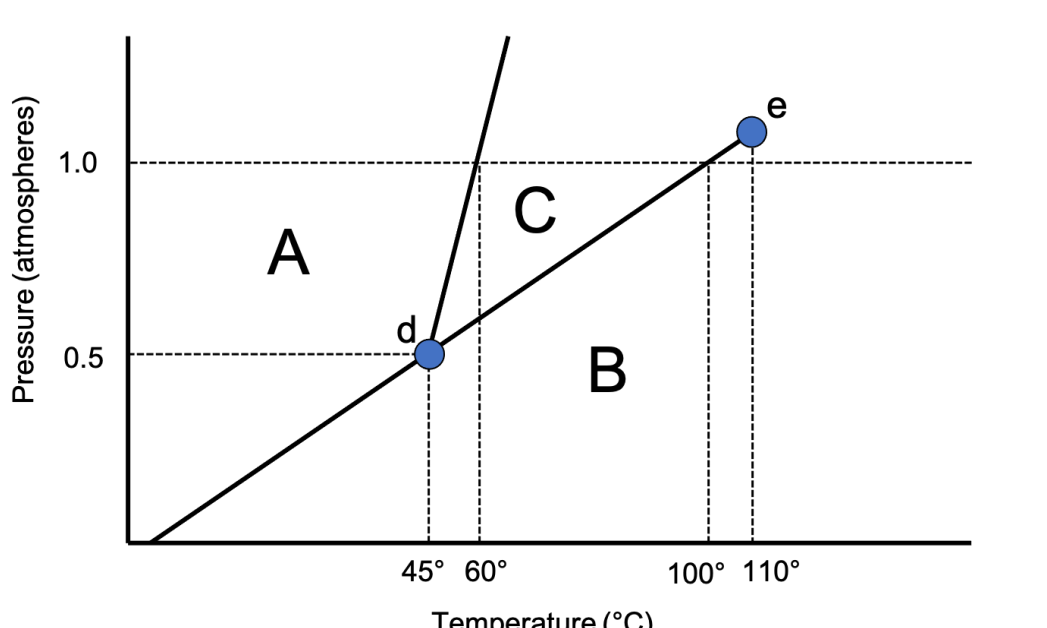
Using this phase diagram, answer the following questions: a. What section represents the solid phase? b. What section represents the gas phase? c. What section represents the liquid phase? d. What letter represents the triple point? e. What letter represents the critical point
Using this phase diagram, answer the following questions:
a. What section represents the solid phase?
b. What section represents the gas phase?
c. What section represents the liquid phase?
d. What letter represents the triple point?
e. What letter represents the critical point
f. What is this substance's normal melting point?
g. What is this substance's normal boiling point?
h. Above what temperature is it impossible to liquify this substance no matter what the pressure?
i. At what temperature and pressure do all three phases coexist?
j. Is the density of the solid greater than or less than the density of the liquid?
k. Would an increase in pressure eventually cause this substance to freeze or melt?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images









