View Policies Current Attempt in Progress Suppose we titrate 20.0 mL of 0.100M HCHO2 (K, = 1.8 × 10-4) with 0.100 M NaOH. Calculate the pH. Before any base is added: pH = i When half of the HCHO, has been neutralized: pH = After a total of 15.0 mL of base has been added: pH = At the equivalence point:
View Policies Current Attempt in Progress Suppose we titrate 20.0 mL of 0.100M HCHO2 (K, = 1.8 × 10-4) with 0.100 M NaOH. Calculate the pH. Before any base is added: pH = i When half of the HCHO, has been neutralized: pH = After a total of 15.0 mL of base has been added: pH = At the equivalence point:
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter16: Reactions Between Acids And Bases
Section: Chapter Questions
Problem 16.51QE
Related questions
Question
Transcribed Image Text:View Policies
Current Attempt in Progress
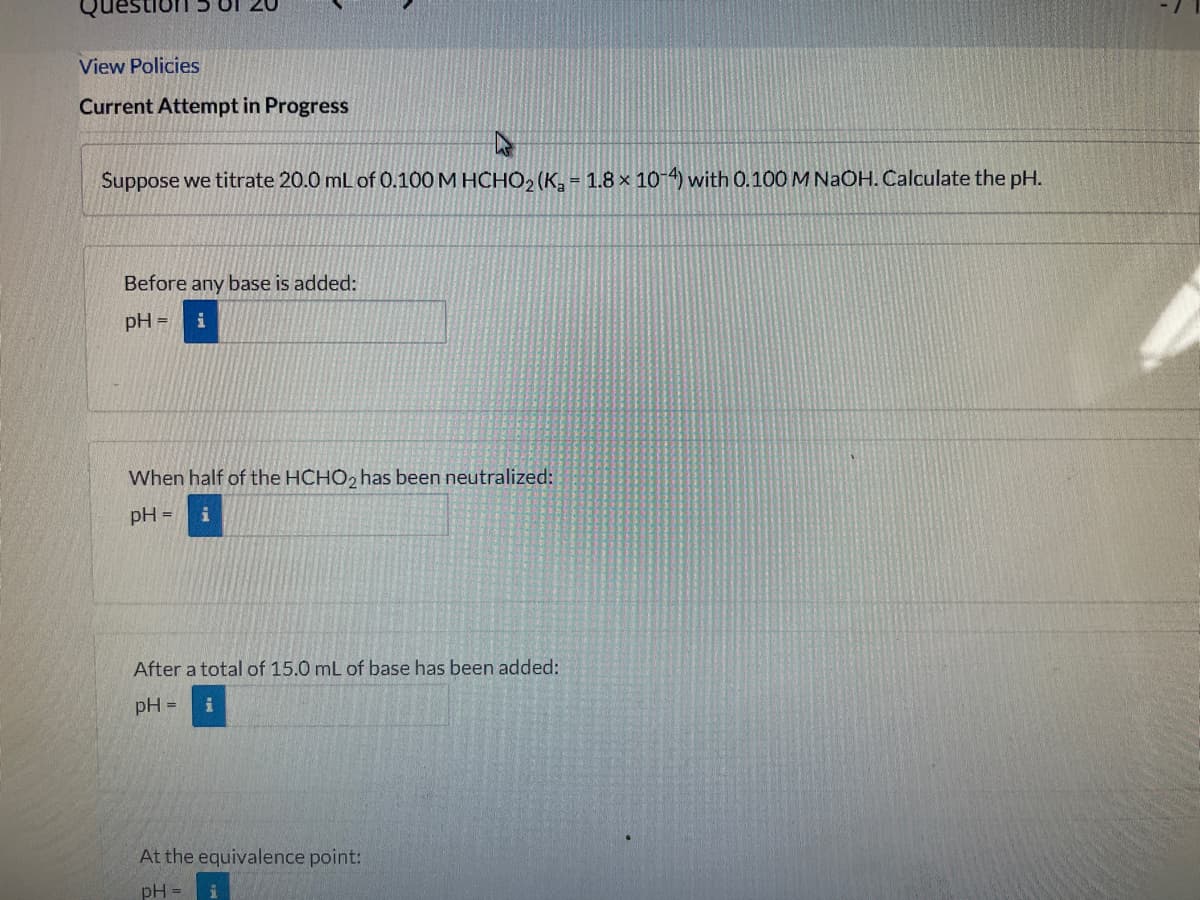
Suppose we titrate 20.0 mL of 0.100M HCHO2 (K, = 1.8 × 10-4) with 0.100 M NaOH. Calculate the pH.
Before any base is added:
pH =
i
When half of the HCHO, has been neutralized:
pH =
After a total of 15.0 mL of base has been added:
pH =
At the equivalence point:
pH =
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


