vo (μM/min) 25 20 15 10 5 0 7 VO (μM/min) VS pH 8 pH 9.5 10.4
Q: Electron Transport Chain, what purpose does it serve in Catabolic pathway?
A: -Anabolism and catabolism are the two main components of metabolism. - The metabolic process known…
Q: 9. Four molecules of ATP are necessary to convert one OAA to PEP. T F
A: OAA is converted to PEP during gluconeogenesis. Enzyme PEP carboxylase catalyzes this reaction.…
Q: For the amino acid tryptophan: Draw its complete protonic equilibria. Indicate the net charge of…
A: The proteins are composed of twenty naturally occurring amino acids. The net charge on an amino acid…
Q: 3 a. b. Name and very briefly describe the four levels of protein structure. Hemoglobin has a…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: write out summary on: Hemoglobin type of Anemia and affects on overall health.
A: Hemoglobin is a blood protein which binds to 4 oxygen molecule to form oxyhemoglobin to supply…
Q: 6. What term is used to describe reactions that generate metabolic intermediates of a pathway? A.…
A: The reactions that generate metabolic intermediates of a pathway is termed as - Answer- B.…
Q: Algae are being studied as a source of lipids to be used as source of biodiesel as a liquid fuel.…
A: Given that algae uses H2O, NH3, CO2 in the presence of light to produce a compound CH1.8O0.5N0.2 and…
Q: what is the dimer of proteins
A: A protein is a biomolecule consisting of many amino acids. It functions as building blocks of the…
Q: Fetal hemoglobin binds 2,3 BPG with greater affinity than adult hemoglobin. True False
A: Growing fetuses' receive oxygen and other nutrients from their moms. Fetal hemoglobin is the…
Q: List the ways in which intact endothelium prevents clotting 6) Know the functions of the coagulation…
A: Coagulation or clotting is the mechanism by which liquid blood changes to a gel like form.…
Q: What is a codon, how many RNA nucleotides are in a codon, How many of these code for amino acids,…
A: Transcription is the process by which the genetic information stored in the DNA is copied onto an…
Q: 1.) How do enzymes affect the proteins that we ate? 2.) How can co-enzymes affect enzyme activity?
A: Enzymes are bio catalysts that work to catalyse a biochemical reaction by decreasing its activation…
Q: Identify the type of glycosidic linkage present in the disaccharide below: O a (1-6) O a (1-4) 03(1…
A: Disaccharides are oligosaccharides composed of two monosaccharide units. The monosaccharide units in…
Q: 15.5 Identify the acid-base conjugate pairs in each of the following reactions: (a) CH₂COO + HCN (b)…
A: An acid is defined as the donator of protons and a base is defined as the acceptor of protons. Acid…
Q: of the gluconeogenesis wing is not true? A. Phosphofructokinase-1 (PFK-1) and…
A: Glycolysis - is a process in which one mole of glucose is partially oxidized into two moles of…
Q: Given the R-groups in the peptide GLY-ASN-THR-LYS-HIS. It would most likely be: a)neither soluble…
A: Different amino acids have different R-groups. The R-groups can be polar or non-polar. Polar…
Q: 2. Aspirin can be absorbed into the blood through the cells lining the stomach and the small…
A: Aspirin is a drug that is used to reduce pain, antiinflammation, and fever. Aspirin is chemically…
Q: Propose a pathway for the following compound to enter gluconeogenesis / glycolysis. In your pathway,…
A: Glycolysis is a collection of 10 enzymatically catalysed reactions that sequentially oxidise a…
Q: Compare extinction coefficients for DNA (1 mg/ml, 260 nm) and the proteins (1 mg/ml, 280 nm).…
A: Introduction There are two types of nucleic acid present in our body, DNA and RNA. DNA acts as a…
Q: The side chain of which residue can be hydrogen bond donor: a. Thr b. Ala c. Ile d. Pro e.…
A: There are twenty standard amino acids that makeup all the proteins present in our body. These twenty…
Q: Question - is if the cells in our bodies were to convert the required energy into our food substance…
A: Energy must be supplied continuously for living things to exist. This energy is employed in part to…
Q: b. You have 100 mLs of a 2 M histidine solution at pH 6.0, how many mls of a 5 M KOH solution must…
A: Dissociation of a weak acid is mathematically described by the Henderson-Hasselbalch equation: pH =…
Q: Explain the role of zinc in biomolecules .
A: Zinc is the micro nutrient and is considered as the third most abundant element in the body. It has…
Q: referred to feedback inhibition as “Goldilocks regulation”. Describe how feedback inhibition keeps…
A: Biochemical pathways and reactions are occurring constantly in cells. These pathways convert the…
Q: Which of the following concerning hemoglobin is true? a) BPG decreases at high altitudes and causes…
A: 1 Hemoglobin is a globular protein, ie it is roughly spherical. It is an oxygen-binding protein that…
Q: Which of the following statement about the enzyme thermodynamics is TRUE? a. Enzymes increase…
A: Introduction Enzymes are known as bio-catalyst. All the metabolic reaction of our body is enzyme…
Q: 5. Bread contains mixture of polypeptides known as gluten. This polypeptide has two types which are…
A: The folding of a polypeptide chain into a three dimensional folded conformation can be divided into…
Q: What typically is found in position X in the molecule below?
A: Nucleotides are molecules that have a nitrogenous base (Adenine, Thymine, Guanine, Cytosine and…
Q: Draw the Citric acid cycle pathway and identify the different types of organic reaction mechanisms.…
A: Citric acid cycle involves 8 biochemical reactions that occur in a cyclic manner. The acetyl CoA…
Q: Describe the specific multi-enzyme example discussed in class of how enzyme activity can be altered…
A: Enzymes are usually comprised of proteins. It work as a catalyst for several biochemical reactions.…
Q: 31. Margarine is made from vegetable oil by a chemical process. Do you suppose this process converts…
A: Margarine is a product derived from vegetable oil, obtained by the hydrogenation,…
Q: ________ is a regulatory mechanism in which the end product of a metabolic pathway inhibits an…
A: Enzymes are biological catalysts. The enzymes, whose ability to catalyze a reaction depends upon…
Q: In affinity chromatography, which of the following strategies is used to elute a GST-POI fusion…
A: GST-POI stands for Glutathione-S-Transferase tagged Protein Of Interest. Glutathione is the…
Q: Explain the role of zinc in biomolecules.
A: Zinc is one of the essential trace elements required for our body. About 10-15mg/dl of Zinc is…
Q: 13. Vitamin H: coenzyme form, functional groups, mechanism of action, biological role, sources,…
A: Introduction: Vitamins are chemical compounds that are found naturally in plants and animals. It is…
Q: d) Ninhydrin is used to turn amino acids in fingerprints purple, make a good way to stain…
A: Note: As per the guidelines, only the first two subparts are to be answered. Kindly resubmit the…
Q: 15. Cellular proteins are oftentimes post-translationally modified. Choose one of the following…
A: Post translational modification are the modifications done in the protein by covalently attaching…
Q: What are the similarities and differences of intermolecular interactions that stabilize secondary…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: The active site _______________. a. is the compound that an enzyme reacts with during the chemical…
A: The active site is that region of an enzyme where substrate molecules bind. The binding of substrate…
Q: In the absence of oxygen is glucose the major source of ATP?
A: It is generally agreed upon that ATP serves as the mediator of energy transfer between anabolism and…
Q: 1. List the 3 final products of glycolysis.
A: Glycolysis breaks down glucose into two molecules of pyruvate that occurs in the cytoplasm and ATP…
Q: Regarding the physical condition (characteristics of the solution/environment) in which an enzyme…
A: Enzymes are large molecular weight proteins that catalyse biochemical reaction. An enzyme's…
Q: The gels we will be using for electrophoresis during the next lab consist of 1% agarose. Suppose you…
A: There are different ways to denote the concentration of a solution. Per cent (w/v) means there is 1…
Q: The side chain of cysteine contains: OA) a hydroxyl group OB) an amine group C) a carboxyl group OD)…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: How does ATP releases the energy used by the Cell/What happens to ATP for the energy release/what…
A: ATP is also known as adenosine triphosphate. ATP is also called the energy currency of the cell. ATP…
Q: Even though HbA is a heterotetramer (not all the subunits are the same), the alpha- and beta-globin…
A: Haemoglobin is a carrier protein that carries oxygen to the cells . It is present in red blood cells…
Q: Give 5 disaccharides, draw its chemical structures, structure of sugar 1, and structure of sugar 2.
A: When two monosaccharides are connected by a glycosidic bond, a disaccharide is formed. Disaccharides…
Q: You have 4 ml of an antigen solution, how would you prepare a 3-fold dilution series such that you…
A: Dilution means decreasing the concentration. 3-fold dilution means decreasing the concentration 3…
Q: Which of the following is not true about a-amino acids? have at least 2 pka values O negatively…
A: The proteins are composed of twenty naturally occurring amino acids. The amino acids can be…
Q: The condensation reaction catalyzed by ß-ketoacyl-ACP synthase synthesizes a four-carbon unit by…
A: Malonyl CoA is formed from carboxylation of Acetyl CoA through an ATP consuming process by biotin…
Step by step
Solved in 2 steps

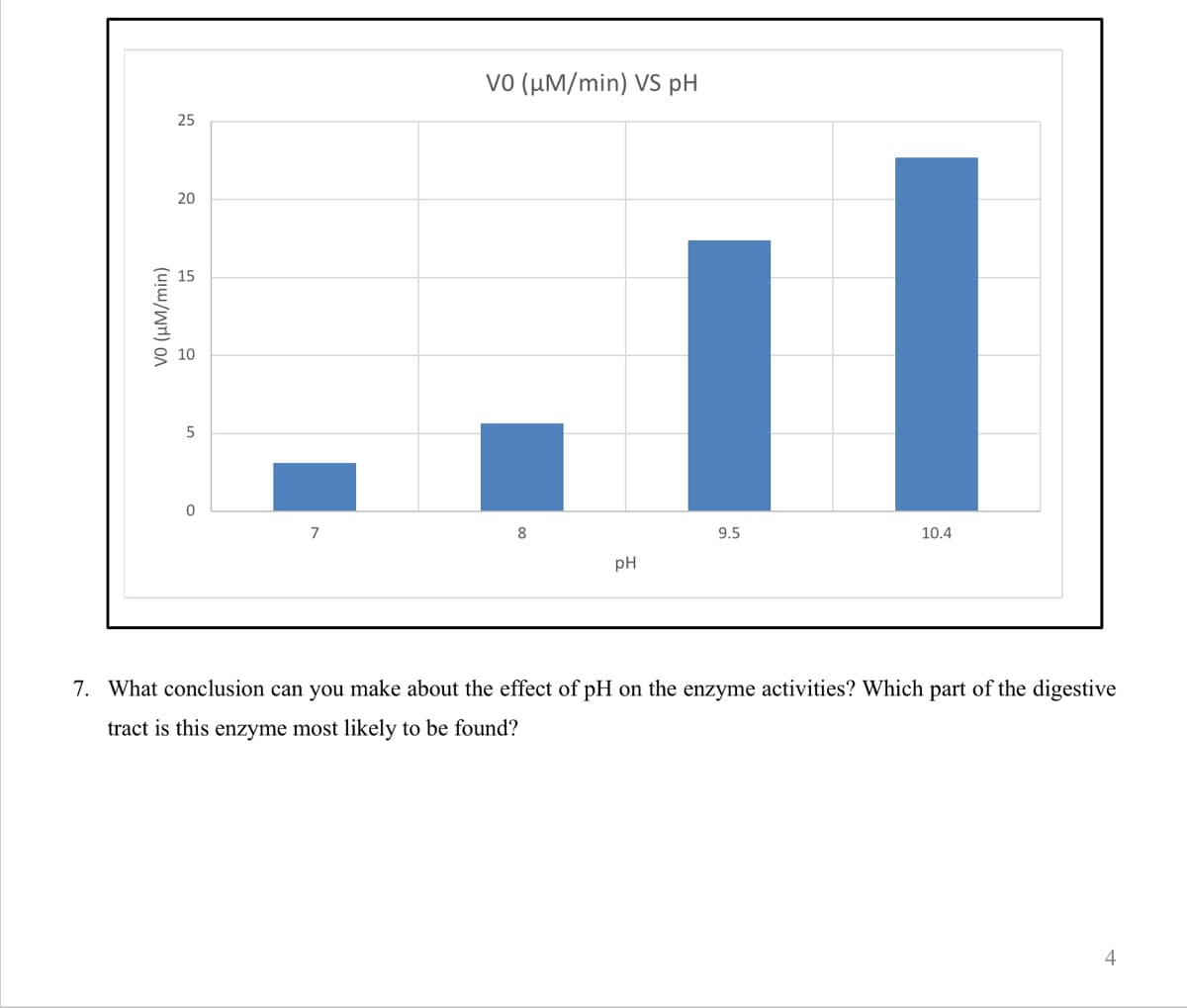
- After the lunch meal, a student felt that the digestion didn't go well. He decided to take some digestive enzyme tablet that helped the digestion. In Michaelis-Menten Equation, which term will be affected? 1.[S] 2. [E]o 3. Kcat ○ 4.Km 05.[1]Estimate the optimum pH for the digestive enzyme trypsin, which hydrolyzes proteins in the small intestines, from the following graph.Why an average meal requires approximately 4 h for complete absorption?
- How will you prepare 750 ml of the digestion buffer. Show all steps and calculations and remember to explain how exactly you will make it up.Interestingly, we can measure metabolism through respiration, with what’s called the respiratory quotient. What is it? What does a decrease in the non-protein respiratory quotient (npQR ) tell us about the severity of liver disease? Is the liver in this case relying more on Glycogen or Fat?Given the nutritional principle of bioavailability, which of the following food combinations would most effectively increase the absorption of non-heme iron?A) Spinach salad with lemon juice dressingB) Whole grain cereal with milkC) Coffee with a bean burritoD) Eggs with a side of whole grain toast
- In a test tube of starch, amylase, and Benedict's reagent, what color would result and does carbohydrate digestion occur? What about starch, water, and Benedict's reagent?At what pH values is pepsin likely to be denatured? Justify your answer.Using the standard curve for the digestion of amylose producing maltose (Figure 13, Workshop 4 manual), determine the amount of maltose produced for each of the conditions (Tubes 2-10). Assume that these values are the amount of product (maltose) produced per unit time (e.g., mM per second) or enzyme velocity Absorbance (545 nm) Maltose (mM/sec) 0 0 0 0.040 0.210 0.330 0.295 0.280 0.202 0.030
- What is the precursor of bile salts and what is their role in the human digestive system?Gastric juice normally has a pH of 2. Given this, what happens to the activity of salivary amylase, a starch-digesting enzyme in saliva, when it arrives in the stomach?Define a kilocalorie (kcal). How is the unit used? How does it relate to a calorie?







