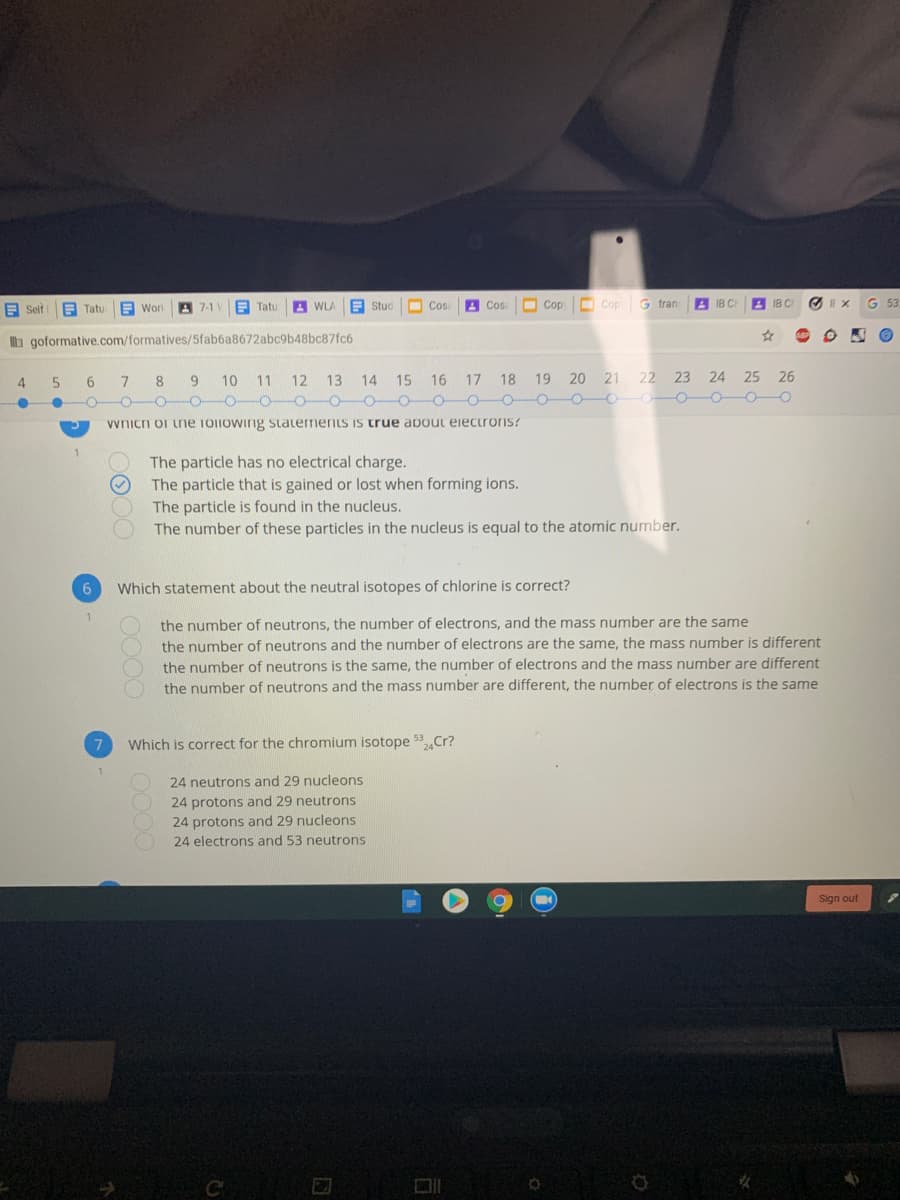
vvnicn or tne Toiiowing statements is true about eiectrois? The particle has no electrical charge. The particle that is gained or lost when forming ions. The particle is found in the nucleus. The number of these particles in the nucleus is equal to the atomic number. Which statement about the neutral isotopes of chlorine is correct? the number of neutrons, the number of electrons, and the mass number are the same the number of neutrons and the number of electrons are the same, the mass number is different the number of neutrons is the same, the number of electrons and the mass number are different the number of neutrons and the mass number are different, the number of electrons is the same 53 Which is correct for the chromium isotope Cr? 24 neutrons and 29 nucleons 24 protons and 29 neutrons 24 protons and 29 nucleons 24 electrons and 53 neutrons
vvnicn or tne Toiiowing statements is true about eiectrois? The particle has no electrical charge. The particle that is gained or lost when forming ions. The particle is found in the nucleus. The number of these particles in the nucleus is equal to the atomic number. Which statement about the neutral isotopes of chlorine is correct? the number of neutrons, the number of electrons, and the mass number are the same the number of neutrons and the number of electrons are the same, the mass number is different the number of neutrons is the same, the number of electrons and the mass number are different the number of neutrons and the mass number are different, the number of electrons is the same 53 Which is correct for the chromium isotope Cr? 24 neutrons and 29 nucleons 24 protons and 29 neutrons 24 protons and 29 nucleons 24 electrons and 53 neutrons
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter1: Basic Concepts Of Chemistry
Section: Chapter Questions
Problem 43GQ: Hexane (C6H14, density = 0.766 g/cm3), perfluoro-hexane (C6F14, density = 1.669 g/cm3), and water...
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
Transcribed Image Text:Selti
E Tatu
E Wori A 7-1 V
E Tatu
4 WLA
E Stud
O Cos
4 Cosa
O Cop D Cop G tran
A IB C
A IB CI O x
G 53
b goformative.com/formatives/5fab6a8672abc9b48bc87fc6
19 20 21 22 23 24 25 26
0-O0- O00 0-00-00 O0 0 000
6 7 8 9 10 11 12 13 14
15
16 17
18
vvnich or ne Toiiowing staterments is true about eiectroris?
The particle has no electrical charge.
The particle that is gained or lost when forming ions.
The particle is found in the nucleus.
The number of these particles in the nucleus is equal to the atomic number.
Which statement about the neutral isotopes of chlorine is correct?
the number of neutrons, the number of electrons, and the mass number are the same
the number of neutrons and the number of electrons are the same, the mass number is different
the number of neutrons is the same, the number of electrons and the mass number are different
the number of neutrons and the mass number are different, the number of electrons is the same
7
Which is correct for the chromium isotope 53Cr?
24 neutrons and 29 nucleons
24 protons and 29 neutrons
24 protons and 29 nucleons
24 electrons and 53 neutrons
Sign out
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co