Would you use acid-catalyzed hydration or oxymercuration/demercuration to form the alcohol product shown? Provide reaction conditions and the structure of the starting alkene. Draw the mechanism of your proposed reaction. OH НО CH3 CH₂ CH3 Propose a synthetic route to make the product from the given starting molecule. now the product after each step you propose. Steps?
Would you use acid-catalyzed hydration or oxymercuration/demercuration to form the alcohol product shown? Provide reaction conditions and the structure of the starting alkene. Draw the mechanism of your proposed reaction. OH НО CH3 CH₂ CH3 Propose a synthetic route to make the product from the given starting molecule. now the product after each step you propose. Steps?
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section: Chapter Questions
Problem 20.42P: Bicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide...
Related questions
Question
Qa 23.
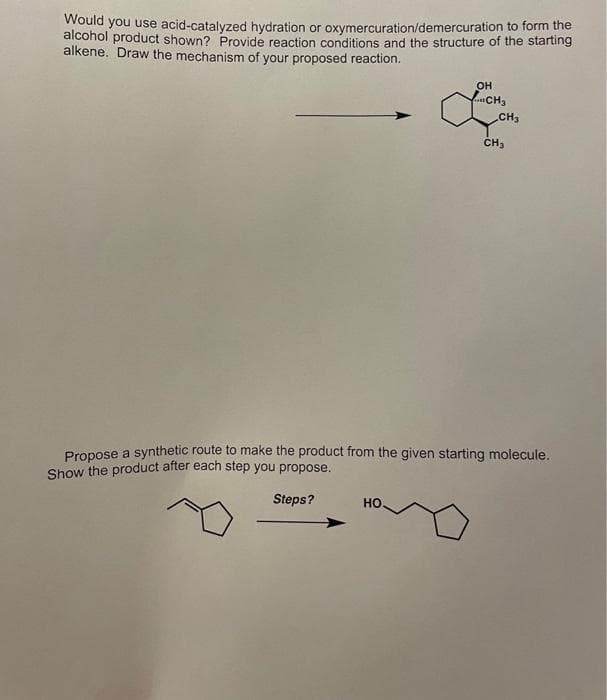
Transcribed Image Text:Would you use acid-catalyzed hydration or oxymercuration/demercuration to form the
alcohol product shown? Provide reaction conditions and the structure of the starting
alkene. Draw the mechanism of your proposed reaction.
OH
...CH3
CH₂
HO.
CH₂
Propose a synthetic route to make the product from the given starting molecule.
Show the product after each step you propose.
Steps?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning