What functional groups are present in the peaks?
Biomedical Instrumentation Systems
1st Edition
ISBN:9781133478294
Author:Chatterjee
Publisher:Chatterjee
Chapter1: Introduction To Biomedical Instrumentation Systems
Section: Chapter Questions
Problem 4Q
Related questions
Question
What
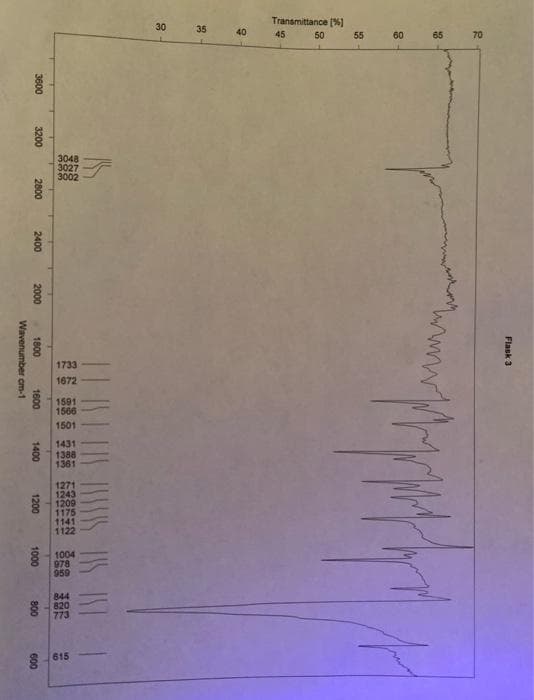
Transcribed Image Text:Transmittance (%)
30
35
40
45
50
55
60
65
70
1733
1672
1566
1501
8615
Flask 3
www.
ISSSS IS IST
3600
3200
2800
2400
2000
1800
1600
1400
1200
1000
800
600
Wavenumber cm-1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you



Case Studies In Health Information Management
Biology
ISBN:
9781337676908
Author:
SCHNERING
Publisher:
Cengage



Case Studies In Health Information Management
Biology
ISBN:
9781337676908
Author:
SCHNERING
Publisher:
Cengage
