Q: hat type of carbocation rearrangement has taken place over the course of the reaction? CI ELOH EtO…
A: Shifting in a chemical reaction is take place because of attain stability.
Q: 10. NBS KOBr (A) . Product (A) is : LOH HO- NH (a) (b) (c) (d) -NH2 - NH2
A: This reaction is Hoffmann Bromamide name reaction and intermediate is 'nitrene'.
Q: 6. perform a retrosynthetic analysis by working backwards two steps in the synthesis below.
A: Retrosynthesis is a technique for solving problems in planning the organic synthesis. It is a…
Q: Which compound will yield a positive result with Salkowski test? * он он он он H;C CH, OCH, но A B…
A: The correct option are as follows:
Q: Which of the indicated sites on the molecule shown is the most basic? (D). NH2 (C) (A) (B)
A: Given : Compound is given with labelled atoms. To find : Atom that is most basic Solution : In…
Q: 4. Give R and S configuration to the following compounds. OH CH CH3 CHa HO
A: Configurations: R S R R
Q: Which of the following reaction sequences will give the meso product? H D DH meso only 1| D, Pd…
A: Given:
Q: What conditions give the transformation shown? H how? (1) Og: (2) ZnHOAC LI/ NH3lia) ob. (1) Brg:…
A: The given reaction is,
Q: 8. Choose the best sequence that would accomplish the following transformation. SO,H 1. X 2. Y Br A.…
A: First benzene reacts with SO3/H2SO4 to give benzene sulphonic acid. This process is called…
Q: Which reaction below give a par of diastereomers and Why? H CH3 CH;COOH А) H CH; HBr B) pегоxides…
A: Diastereomers are stereoisomers that are not mirror images of one another and are non-superimposable…
Q: Teken een geldige resona volgende bensielkation Draw one valid resonance intermediate for the…
A: Benzylic carbocation is stabilized through resonance.
Q: 34. Select the best synthetic sequence to accomplish the transformation shown. Br A. 1. NaOEt; 2.…
A:
Q: 1. Draw a plausible mechanism for the following transformation. H H2SO4 N° + 2H20 + NH2NH2 H
A:
Q: :0: CH3OH2* protonation H3C CH3OH nucleophilic addition CH3 CH3OH H Drawing :OCH, deprotonatio =D1…
A:
Q: Select the correct product for the following transformation: Br. Br NaCN ? DMSO NC A B с D A 'Br Br.…
A: Note : DMSO polar aprotic solvent and the alkyl halide is primary alkyl bromide. Therefore the…
Q: d) What is the IHD (index of hydrogen deficiency) of the following compound? (don't calculate the…
A: d) The hydrogen deficiency index can be calculated by the number of the double bonds (pi-bonds) and…
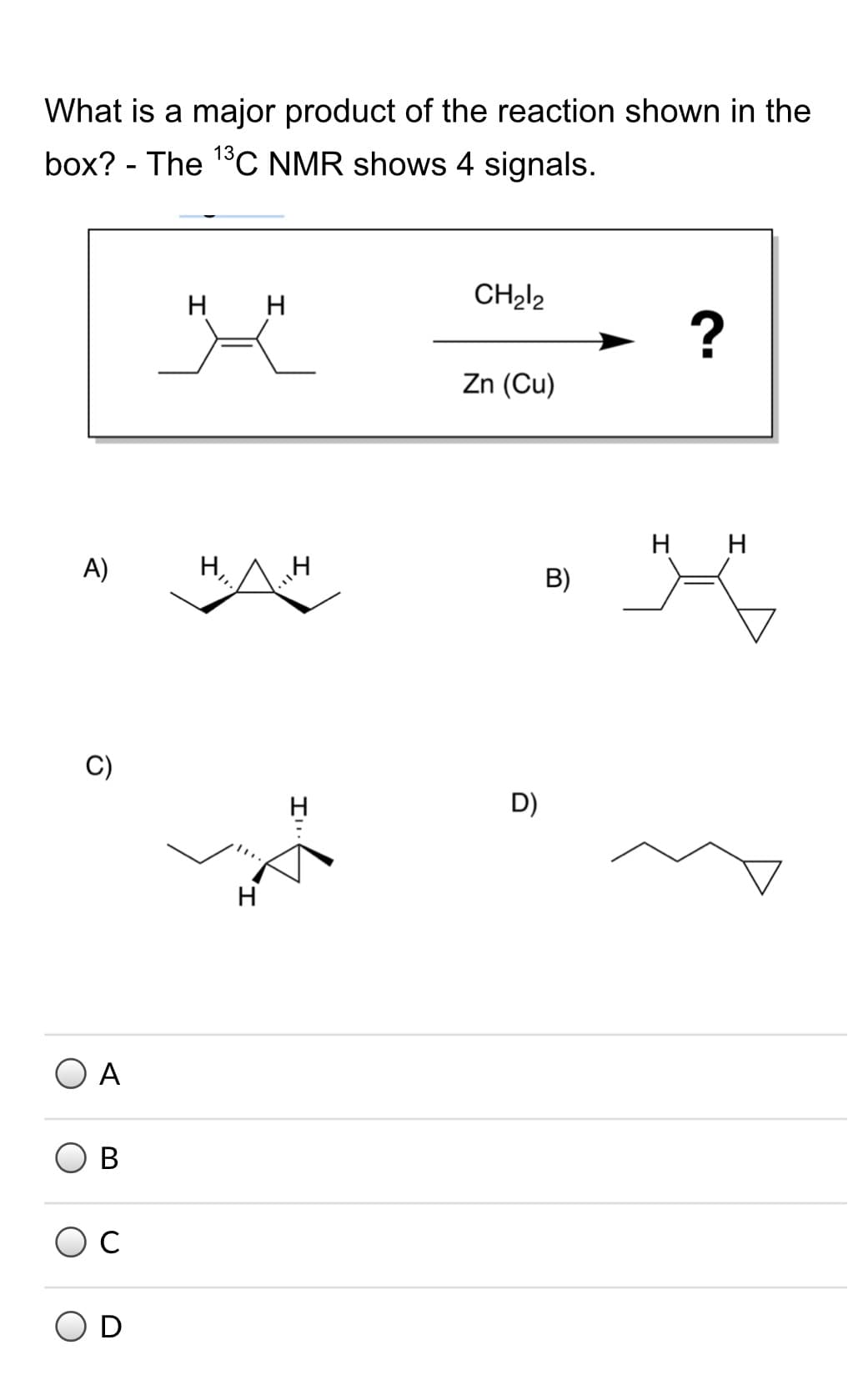
Q: 18. What is a major product of the reaction shown in the box? The 1°C NMR shows 4 signals. H H CH212…
A: To answer this question , we would first complete the reaction from given reactant and reagents.…
Q: 17. Draw the mechanism for the following SN1 processes. OH Br OH HBr HBr Br (a) (Ь) OEt H20 ELOH (c)…
A: The mechanism of given SN1 reactions are given below -
Q: 21. What is a major product of the reaction shown in the box? Br Pd-cat. ? Base H NMR of the major…
A:
Q: Draw both diastereomers of 2-bromo-3-chlorobutane by editing the two drawings of…
A:
Q: To which compound is the addition of HBr more highly regioselective (without any shift) and what…
A:
Q: What product is formed in the Suzuki coupling of A and B? This reaction was a key step in the…
A:
Q: 1. HSCH,CH,CH,SH H. BF3 OEt, 2. п-BuLi 3. Br 4. HgCl, /H,O THF (solvent) 5. heat [3,3] sigmatropic…
A: Step 1: Protecting the carbonyl group with the HSCH2CH2CH2SH in presence of BF3OEt2 as shown below.…
Q: -NH2 is a(n) O weakly activating group. O weakly deactivating group O ortho/para director. O meta…
A: Since in -NH2 group we have lone pair electron in N and the substitutes attached to N are H only…
Q: 23. What is a major product of the reaction in the box? 1. OsO4 ? 2. NaHSO3, H2O 1°C NMR of the…
A:
Q: 6. Propose how to carry out the following reactions. (13.36,13.37) CN Enantiomer CN
A: Solution 13.36) Enantiomers area unit chemical isomers that area unit non-superimposable mirror…
Q: CH30
A:
Q: A trisubstituted be bromine and two methoxy substituents exhibits three aromatic resonances at 8 =…
A:
Q: What arethe reagents needed to complete this reaction? a. Hz/ Lindlar's cataly st b. Li, E+NH2,-78°…
A: Welcome to bartleby ! We have to tell the appropriate reagent for the above transformation .
Q: 1. Which of the following is a major product of the reaction shown in the box? – 13C NMR: 9 signals.…
A: NMR is branch of chemistry in which we deal with shielding and deshielding effect on nucleus. It…
Q: ·BH 3. H₂0₂ 0.00 Predict the Product with Stereochemistry
A:
Q: 2 Conseeutive 3,3- sigmatropic rearrangements 2.
A:
Q: Two 2-step sequences are shown below to make m-bromoacetophenone. Which one will work better?…
A:
Q: 2. Synthesize the target compounds (C1-5) by matching the following starting materials with the…
A: The given compound is synthesized by the given carbonyl compound and the carboxylic acid. The…
Q: How many types of non-equivalent protons are on fluorobenzene? 2 5 4 3…
A: The protons present in the same type of environment is called an equivalent proton. When the…
Q: D5D Which compound will form the most stable cyanohydrin? (Hint: Cyanohydrin formation exhibits ap…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: 1. Draw the intermediates A-C for reactions a and b. Please show the appropriate stereochemistry…
A:
Q: 6. Which of the following is a major product of the reaction sequence shown in the box? H2NNH2 КОН ?…
A:
Q: 3. (a) The structures of [14]-annulene and [18]-annulene are as shown below. H H. [14]-Annulene…
A: Aromatic compounds are those compounds which obey Huckel's rule and has (4n+2) pie electrons.
Q: 3. Rearrange the molecules from strongest IMFS to weakest. C,H0 C,H, CH C,H
A: Answer:- This question is answered by using the simple concept of intermolecular forces (IMF) of…
Q: What is the product of the following reaction: N- Br, / FeBr3 ?? Br H O Br H. Br
A: Br2/FeBr3 used forFeBr3 acts as a Lewis acid in this reaction.
Q: Provide the major product in each box for the multistep synthesis below. Mg0 Br Et,0 2. aq NH CI 55…
A: Alkyl magnesium halide is known as a Grignard reagent. The general formula of the Grignard reagent…
Q: 1. Draw the resulting radical (phenyl, C6H5) after the H abstraction.
A: Benzene is an aromatic organic compound. Benzene obyes Huckel's rule. The molecular formula of…
Q: What product is formed in the Suzuki coupling of A and B? This reaction was a key step in the…
A: In Suzuki reaction, an organoboron compound is reacted with an aryl (or alkenyl) halide in the…
Q: Indicate the reagent(s) needed to carry out the following transformations. (Ctrl) с) CHз-СH2-CH2-ОН…
A: In the first question, there is a substitution that takes place i.e substitution of one nucleophile…
Q: Br A Br B 1. AICI 3, 2. Zn(Hg), HCI 3. Br2, FeBr3 Br ? D Br E Br
A: In the Friedel-Crafts acylation reaction, an acyl group is attached to an aromatic ring. This is…
Step by step
Solved in 4 steps with 3 images

- Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Draw a reaction scheme of the deprotonation of a benzylic proton with LiN(SiMe3)2Reaction of p-cresol with two equivalents of 2-methylprop-1-ene affordsBHT, a preservative with molecular formula C15H24O. BHT gives thefollowing 1H NMR spectral data: 1.4 (singlet, 18 H), 2.27 (singlet, 3 H), 5.0(singlet, 1 H), and 7.0 (singlet, 2 H) ppm. What is the structure of BHT?Draw a stepwise mechanism illustrating how it is formed.
- select the most appropriate reagent(s) to effect the change. K2Cr2O7, H+ H2, Pd 1. Disiamylborane, 2. HO–, H2O, H2O2 NaOCl H2SO4, HgSO4Which of the following reactions result in a positive ∆ Ssys? A. Pb(NO3)2 (aq) +KCl (aq) ---> PbCl2 (s)+ KNO3 (aq) B. HCl (g) + H2O (l) ---> HCl (aq) C. H2 (g) + I2 (g) ---> 2HI (g) D. 2H2O (g) ---> 2H2 (g) + O2 (g) E. H2O (g) ---> H2O (l)Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents? a) (CH3)2CU-Li+, then H3O+ b) LiA1H4, then H3O+ c) CH3NH2 d) C6H5MgBr, then H3O+ e) H2/Pd f) CrO3, H3O+ g) (C6H5)3 P+C- HCH3 h) HOCH2CH2OH, HC1
- (CH3)3CO- is a base that can be made from (CH3)3OH (pKa = 18). Using curved arrow formalism, draw a reaction of this base reacting with NH3 (pKa = 36). Show the products. Which way will be equilibrium lie? Explain.Which reagent(s) would accomplish the following reaction? 1) LiAlH4 and 2) H2O 1) CH3MgBr and 2) H2O H2O, H+ NaH NaOH, H2OCH3)3CO- is a base that can be made from (CH3)3COH (pKa = 18). Using curved arrow formalism, draw a reaction of this base reacting with NH3 (pKa = 36). Show the products. Which way will be equilibrium lie?

