What is the calculated value of the cell potential at 298K for an electrochemical cell with the following reaction, when the Hg²* concentration is 1.24×104M and the Mn2* concentration is 1.26 M? Hg**(aq) + Mn(s)–Hg(1) + Mn²*(aq) Answer: |v The cell reaction as written above is spontaneous for the concentrations given:
What is the calculated value of the cell potential at 298K for an electrochemical cell with the following reaction, when the Hg²* concentration is 1.24×104M and the Mn2* concentration is 1.26 M? Hg**(aq) + Mn(s)–Hg(1) + Mn²*(aq) Answer: |v The cell reaction as written above is spontaneous for the concentrations given:
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter20: Electrochemistry
Section20.1: Voltaic Cells
Problem 13SSC
Related questions
Question
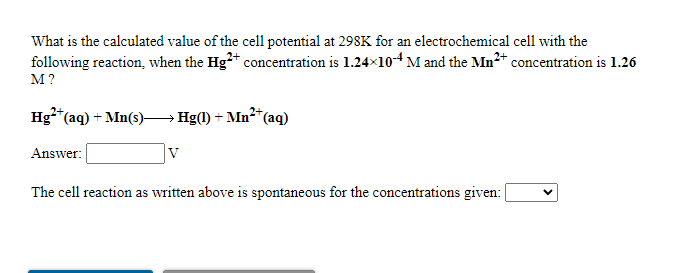
Transcribed Image Text:What is the calculated value of the cell potential at 298K for an electrochemical cell with the
following reaction, when the Hg** concentration is 1.24x104 M and the Mn* concentration is 1.26
M?
Hg**(aq) + Mn(s)→Hg(1) + Mn²*(aq)
Answer:
The cell reaction as written above is spontaneous for the concentrations given:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
