Q: if the possible molecular formula is C8H7ClO what is the structure consistent with all the data
A: Given that, the molecular formula of a compound is C8H7ClO. Also, the 1H NMR spectrum and the mass…
Q: Propose an efficient synthesis the following transformation: (a) The transformation above can be…
A:
Q: 3.1 Which functional group is not found in oxycodone? Lo OH H oxycodone (a) ketone (b) ether (c)…
A:
Q: I: H₂S(aq) + Cl(aq) HCl(aq) + HS (aq) II: NH4 (aq) + HCO3(aq) H₂CO3(aq) + NH3(aq) A) Only I B) Only…
A: Given reactions are : I : H2S (aq) + Cl- (aq) <--------> HCl (aq) + HS- (aq) II. NH4+ (aq) +…
Q: Treatment of a 0.2500-g sample of impure potassium chloride, KCl (74.551 g/mol) with an excess of…
A:
Q: Chemistry Use both a Q-test and Grubb's test on the following data to see if the lowest value can be…
A: The questionable value is the lowest number which is 34. We need the mean(average) and standard…
Q: Shown below is a possible synthesis of the target molecule, starting with a substrate containing two…
A:
Q: Which do you expect to have a more intense and concentrated “hot spot” of negative charge: methoxide…
A:
Q: Briefly explain the techniques of catalyst characterization: c) FT-IR of solids with pyridine…
A: Infrared spectroscopy is a technique which deals with the analysis of a molecule when it reacts with…
Q: Fill out this table, involving the compounds which will be used in this lab: Acetominophen Aspirin…
A:
Q: Consider the following equilibrium for which H = -114.44: 4 HCl(g) + O2(g) 2 Cl2(g) + 2 H2O(g)…
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on…
Q: Calculate the wavelength (in nm) of a light that shines on the surface of a piece of metal with the…
A:
Q: You are performing an experiment that involves the use of atleast 5 test tubes. Which of the…
A: Given that you are performing an experiment that involves the use of at least 5 test tubes. We have…
Q: draw resonance in boxes
A: Resonance →Delocalization of electrons in a molecule Resonance structures contain same number of…
Q: 3) Write the IUPAC name for the following compound. IUPAC name: CI
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Which is a stronger base S^2- or Se^2-? Explain your answer. Please provide answer correctly…
A: Given -> S2- & Se2- -> order of Basicity of conjugate base is same as order of basicity of…
Q: Calculate the mass in grams of each sample 3.9×1022 SO2 molecules to g Express your answer using…
A:
Q: If a solution of dissolved acid has pH = 2.5 and the pka for the acid chemical is K 4.3 and you…
A:
Q: To make 1.50 L of 0.100 M NaCl from a 2.96 M stock solution, you would need to take mL of the stock…
A:
Q: A. Isomer A has only primary
A: Organic compounds are made up of hydrogen and carbon atom. These organic compounds are called…
Q: Methyl group V CH3 -I I N=1 1+1=2 X1H, d Ketone Ketone 1 H 31 symmetric 1H N=4 4+1=5 quintet. Methyl…
A:
Q: Determine the number of moles of HH in each sample. 4.70 mol H2O to mol
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: When comparing two compounds, the more acidic compound will have a lower; lower lower, higher…
A: According to Arrhenius concept, an acid is a substance i.e. when dissolved in water gives H+ ions .…
Q: Order the following solutions from lowest to highest pH: 1. 0.10 M NACIO II. 0.10 M KBr III. 0.10 M…
A: pH is the measure of acidity. It is obtained by negative logarithm of concentration of H+
Q: please help with this question : The research process in which new data will be collected and…
A: A research process can be either primary, secondary, tertiary, or cross-sectional research which…
Q: Can someone check my answers and see if they are correct?
A: The stationary phase (-SO3-) is negatively charged (Cation-exchange chromatography). Since the…
Q: #1: line above the d 7. Р P P Р M. Ann P
A: In an atom, there is nucleus at the centre and electrons are revolving around it. Electronic…
Q: How many double bonds are present in Blue No. 1? (Include the S=O double bonds).
A:
Q: A certain first-order reaction is 33% complete in 55 s. What are the values of the rate constant and…
A:
Q: 2. Given the reactions (1), (2) and (3) below determine the standard enthalpy of formation (AH,) of…
A:
Q: Can the mirror image of bromochloromethane (CH2BrCl) be superimposed on the original? (chirality)
A:
Q: During a routine check of the fluoride content of Gotham City's water supply, the given results were…
A: The replicate concentration values are: 0.815 mg/L, 0.789 mg/L, 0.811 mg/L, 0.789 mg/L, and 0.815…
Q: Q40: Draw the pairs of mirror images using wedges and dashed wedges. mirror image of butan-2-ol…
A:
Q: be + NaOH ?
A:
Q: What is the symbol of potassium
A: Given, The symbol of potassium is:
Q: For the reaction shown, calculate how many moles of NH3NH3 form when each amount of reactant…
A:
Q: How many atoms are in 159 g of calcium? 159 g = 4.13 Incorrect atoms
A:
Q: What is the value of the H—C—H bond angles? (remember VSEPR theory
A: VSEPR (Valence Shell Electron Pair Repulsion) theory is a model which gave the structural…
Q: If the decomposition of a chemical is monitored at 25°C and the initial concentration is 3.1 x10-2…
A: First-order reaction: The rate of the first-order reaction depends only on one reactant. According…
Q: Use the References to access important values if needed for this question. The compound methanol,…
A: Given, Reaction : 2CH4O(I) +3O2(g) →2 CO2(g) +4H2O(g)
Q: Chemistry When 248. mg of a certain molecular compound X are dissolved in 65.0 g of benzene (CH),…
A:
Q: Mercury, essential lemon oil and water are liquids that do not mix with each other. The density of…
A: The density of mercury is 13.6 g/mL the density of lemon oil is 0.85 g/mL. the density of water is…
Q: The aurocyanide ion, [Au(CN)₂]¯ is a coordination complex of gold with two CN™ ligands. When silver…
A: Here we have to predict whether the complex anion Ag(CN)21- form compound with potassium or with…
Q: ou have an aqueous solution of Ca(NO3)2 with a molality of 7.26 mol/kg and a density of 1.4963 g/mL.…
A: The mole of solute present in a one-liter solution is known as the molarity of the solution. The…
Q: Does chloromethane contain a plane of symmetry? Is it a chiral?
A: First of all it needs to know about the chiral centre. What is Chiral? Let us see it in next…
Q: An evaporator is used to concentrate cane juice. 10,000 g of juice containing 38% mass/mass of sugar…
A: Given -> Initial mass of juice = 10000 gm Initial concentration (m/m) of sugar = 38% Final…
Q: The following reaction has been proposed to occur in two elementary steps. Overall Reaction: NO₂(g)…
A: Chemical kinetics can be defined as the branch of the chemistry that deals with rates of chemical…
Q: 1. Draw the mechanism and predict the major product for each of the following reactions to NH₂ OH…
A:
Q: absolute configurations of the two stereogenic centres (i) and (ii) in the structure below…
A:
Q: Describe and write about the history of the atomic theory. Compare the five theories.
A: Atomic theory: The theory that tells about the structure of the atom is named atomic theory. Atomic…
what is the chirality centeris an R or S absolute configuration?
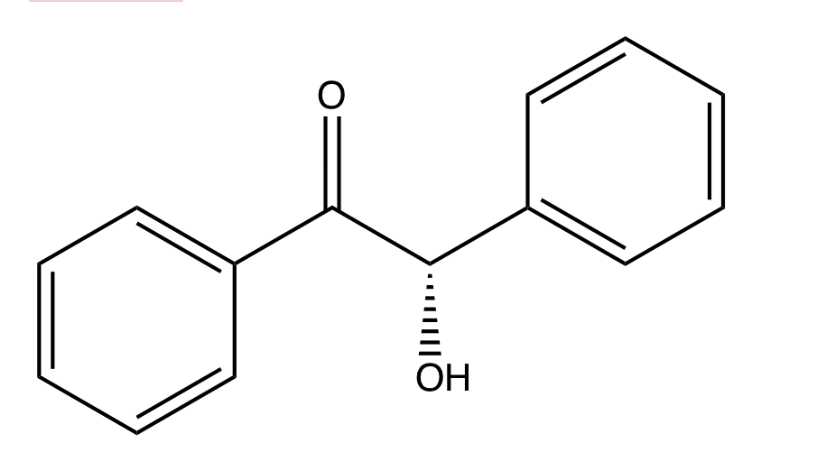
Step by step
Solved in 3 steps with 1 images


