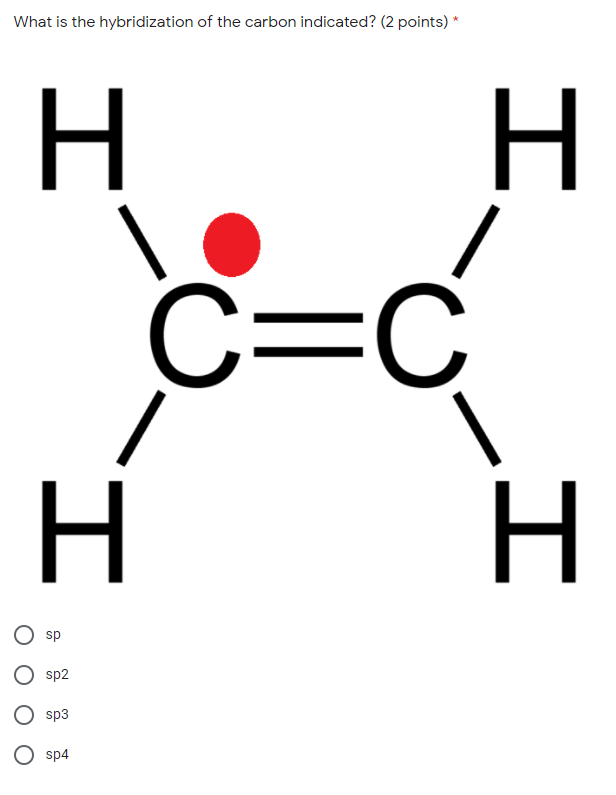
Q: which molecular geometry can form from an sp^3d hybridized center? check all that appy.
A: Molecular geometry defines how atoms of a molecule are arranged in 3 dimesnsion.
Q: 11. What is the hybridization state of the carbon (marked as I) atom in the following compound? H.…
A: Interpretation: The hybridization state of the carbon (marked as I) atom in the given compound is to…
Q: 5. Which of the following molecules and ions would have the same three-dimensional structure as…
A: The molecules having same structure as that of BrF3 are given in step 2
Q: 6. Give the molecular formula for the molecule below NH Draw in all lone-pair electrons on the…
A: Hybridization is the mixing of orbitals of similar energy to get the identical orbitals of same…
Q: why is the answer BCDA? Rank the following molecules in order of increasing dipole moment, from…
A:
Q: Anthracene is a yellow, crystalline solid found in coal tar. Complete the structure for anthracene,…
A: We know that Antharacene is a yellow, crystalline solid found in coal tar.
Q: sk 4. Assign D and L configuration for the following molecule.
A: The isomers are the ones which have same molecular formula but different arrangement of atoms in a…
Q: The C-C bond angles for the ring in M2 is? 60º, 109.5º, 120º or 180º
A:
Q: 1. a) Determine the bond order of emphasized atoms in the following molecules and then ran! them in…
A: Since part (b) of the question is incomplete, we will solve part (a) for you. To get the remaining…
Q: Write down all the symmetry elements associated with the following molecules and then use the flow…
A: The structure of Benzotrifuroxan is as follows:
Q: 12) a. Draw the pi orbitals for molecule 8 for the bonds indicated in blue (and bolded). F N-…
A: Interpretation- To draw the pi-orbital for the blue and bolded part of molecule-8 which is given…
Q: practice q 9 9. For each of the following reactions, assign with a check in the box which of these…
A:
Q: 11.48 Tryptophan is one of the amino acids found in proteins: H H H N-H нн H- :0: H (a) What is the…
A: Hybridisation is the concept of mixing of atomic orbitals to form bonds between atoms. sp3…
Q: C) What is the hybridization of the indicated atom? Circle your answer. sp sp? sp3 D) What is the…
A: To Solve this problem we have to decide the hybridization of the indicated atoms in the given…
Q: Which statement regarding the hybridisation of carbon in the following compound is correct? Select…
A: Given molecule,
Q: 35. Y =[co, – a//3) ¢,] represents normalised a molecular orbital constructed from two different…
A:
Q: How many TT electrons are in the delocalized TT-system of this molecule? 6. 10 12
A: Given the molecular structure. From this, it is clear that there are 3 double bonds in it.
Q: 5. Which of the following is the correct depiction of induction for a C-F bond? ctF C-F C-F II II IV
A: We Know that, Electronegativity of F = 3.98 Electronegativity of C = 2.55
Q: Aflatoxin B1 is a carcinogen that damages DNA molecules. Given the molecular structure: Fill in the…
A:
Q: For the molecule below: CH3 H H Cq is sp3 hybridized, N is sp² hybridized and O is sp2 hybridized. C…
A:
Q: 7. What is the hybridization state of the indicated atoms in the following compound? IV II III A) I-…
A:
Q: Would you expect a C¬C s bond formed by sp2–sp2 overlap to be stronger or weaker than a C¬C s bond…
A: The strength of the C-C bond in which both carbons are sp2 hybridized and that of the strength of…
Q: Assign Re and Si face to an SP2 hybridized carbon (C=O or C=C) Can you explain how to get it and…
A: Re and Si face are face of attack by reagent.
Q: app.101edu.co Determine the hybridization of the indicated carbon atom in the compound shown here.…
A: Hybridization of carbon (C) in hydrocarbons: a) Alkane -C-C- in this case all bonds are single and…
Q: B A) sp B) sp C) sp' D) sp'd E) s'p 7. What is the hybridization state of the indicated atoms in the…
A:
Q: 1.43 For each type of bond below, determine the direction of the expected dipole moment: а. С—О b.…
A:
Q: B. What is the angle between bonds in each of the following? Angle H18 H H H-clc-c-H H H H 24 18 23…
A: Bond angle is defined as the the angle formed between two covalent bond. Rule- Determine…
Q: Q 1.4: The following compound has a total of. sp-hybridized atoms and sp3-hybridized atoms. CH3-C…
A: Carbon having two sigma bonds are sp hybridized and having 4 sigma bonds are sp3 hybridized.
Q: Be sure to answer all parts. Analyze the following image: H. N. H. H. Select the sp2-hybridized C…
A:
Q: What is the hybridization of the central atom? :F- -Xe- -F: Select one: sp3 sp O sp? O sp³d? sp³d…
A: In the hybridization of xenon difluoride, Xenon (Xe) is the central atom. The ground state…
Q: Which molecule below contains an atom that is sp2 hybridized? Select one: O a. HC = CH O b. H3C-CH3…
A: (a) Wrong, It is an alkyne and both the carbons are sp hybridized. (b) Wrong, it is an alkane and…
Q: In which MO is the overlap of atomic orbitals greater, a s2por a p2p?
A: According to the molecular orbital theory,molecular orbitals are formed by the linear combination of…
Q: 1.67 The indicated o bond results from the overlap of which orbitals? HHH H-C C%=C-H a. sp²-sp2 b.…
A: When carbon is attached with four diffarent atoms like H and other C then that centre is Sp3…
Q: 5. (2 points) Consider ethylene, and acetylene ethylene acetylene (a) Draw three-dimensional…
A: In first part of question we are asked to draw the 3-D sketch of Acetylene and ethylene and label…
Q: How many Cs are sp2-hybridized in the molecule shown below? (kindly show explanation or…
A: During the orbital hybridization, the mixing of atomic orbitals takes place to form new orbitals…
Q:
A: The carbon atom containing only sigma bonds is sp3 hybridized, the carbon atom containing one pi…
Q: 1.40 What kind of hybridization do you expect for each carbon atom in the following molecules? (a) H…
A: The most of atoms combine their s and p atomic orbitals to create the most stable molecular…
Q: For the molecule shown below, identify the hybridization on the C atom with the double bonded oxygen…
A: For the molecule shown below, identify the hybridization on the C atom with the double bonded oxygen…
Q: Select the correct statement(s) from the following molecule: но н н H-C-C-C=C-H 3 2 1 H. I. There…
A: Sigma bond is formed by head on overlapping between the orbitals whereas pi bond is formed by side…
Q: Based on the MO diagram provided, what is the bond order of an 02 molecule? ? Submit Request Answer…
A:
Q: What are the x and n values for XeOxFy" molecule to have molecular geometry of tetrahedral and n =…
A:
Q: 5. Fine the species with a maximum dipole among the following | a) NF3 c) NH3 e) CH4 b) CIF; d) CO2
A:
Q: What is the hybridization at each carbon atom in the molecule? Drag the appropriate labels to their…
A: Hybridization is the mixing of orbitals of similar energy to get the identical orbitals of same…
Q: 11. a. Draw the MO diagram of N-. b. Give the MO ground state electron configuration of N-. c. What…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 11 of 17 > If the symbol X represents a central atom, Y represents outer atoms, and Z represents…
A:
Q: 47) Combination of two p-atomic orbitals give two molecular orbitals. Show the possible two…
A:
Q: 1) Below is the drug Brucine, answer the following questions about Brucine. c) Assign the…
A: a) The brucine molecules contain the chiral carbons and it does not contain any plane of symmetry or…
Q: What direction does the dipole moment for the following molecule point in? (A) H = 0 (B) (C) (D)…
A: Dipole Moment Dipole moment is defined as the product of the two charges and the distance by which…
Q: The absolute configuration of C, and C, in the following compound is HO H O H3C HO. 4 Br H. (a) 2R,…
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority.2) If first atom is…
Q: Which of the following molecules are paramagnetic: H2+1, H2, H2-1, H2-2? Group of answer choices H2,…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Which of the following molecules are paramagnetic: H2+1, H2, H2-1, H2-2? Group of answer choices H2, H2-1, H2-2 H2+1, H2-2 H2-1, H2-2 H2+1, H2, H2-1, H2-2 H2, H2-2 H2+1, H2-1 H2+1, H2, H2-2 H2, H2-1 H2+1, H2-1, H2-2 H2+1, H2 H2+1, H2, H2-1CBrIFCl has a tetrahedral shape. Show the final structure after the operations C4(z) σh C2(z)(anticlockwise rotation)Identify the hybridization of the labeled carbons in the molecule Remdisivir. Answer all labels 1 to 10.
- Draw the 4dxy orbital on a x,y axesFor the molecule shown below, identify the hybridization on the C atom with the double bonded oxygen and the approximate bond angle labeled beta: Group of answer choices hybridization: sp2 bond angle: 120° hybridization: sp3 bond angle: 109.5° hybridization: sp3d bond angle: 90° hybridization: sp3 bond angle: 120°which molecular geometry can form from an sp^3d hybridized center? check all that appy.
- I just need help with e and f. Please I have a-c have been answered.Answer the following questions and use a MO Diagram: a. What is the bond order for a CF bond? b. Is the bond stronger or weaker in CF+? c. is the bond stronger or weaker in CF-? d. Propose another bond with the same bond order as CF. e. What would be the bond order of a Ne2 bond?Use VSEPR to predict the three indicated bond angles. All of your options given below are in degrees of angle and are listed in the order a/b/c Group of answer choices 109.5/90/109.5 109.5/120/109.5 109.5/120/90 90/90/90 120/120/90Consider the following orbital representation of HCCH (ethyne). a. Answer the same three questions (a-c) from the previous exercise. b. Label each CH bond in the drawing above. c. What is the total number of a bonds found in ethyne?…. bonds? d. How many p orbitals are there on a single carbon of ethyne? e. How many hybrid orbitals are there on a single carbon of ethyne?

