Q: Show that for a first-order reaction R → P, the concentration of product can be represented as a…
A: To Show that for a first-order reaction RPthe concentration of product can be represented as a…
Q: 1. The three properties of chemical systems at equilibrium are: b. 2. What is meant by "the…
A: 1: We have to write three properties of system at equilibrium2: We have to write the concept behind…
Q: Two drugs have a selectivity factor a = 1.08 and capacity factors k, = 5.0 and k₂= 5.4. If the plate…
A:
Q: 1. Find the molal concentration of a solution containing 8 moles of Sodium Hydroxide and 32 Kg of…
A: Moles of Sodium Hydroxide (NaOH) = 8 molesMass of water = 32 kgTo find the molal concentration of…
Q: 9. What is the single oxidation number of N in NO₂ (aq)? a. +2 b. -2 C. +3 d. -3 e. -6 10. Balance…
A:
Q: N + KOH + Br₂
A: An amide is treated with bromine in an alkali solution in Hoffmann's bromamide reaction, which…
Q: What mass in grams of precipitate theoretically forms when 0.500L of 0.100M calcium hydroxide is…
A:
Q: o Predict the two major organic products of the reaction. (HI behaves as an HX reagent.) H₂C ΤΟ +…
A: Alkenes react with HX to form alkyl halides.Addition of…
Q: What is the IUPAC name of the following compound? ball & stick + labels
A: In ball - stick model, the general trend is-Red balls - oxygen atomGrey balls- carbon atomWhite…
Q: 1. In which example does oxygen have a 1- charge? a. Sc₂03 b. Li₂O c. H₂O2 d. CO2
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: مه h. H H3C Br H CH3 Br CH3 CH3 and and H H3C H Br Br CH3 CH3 CH3
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: Without repetitions, how many possible monochloroalkanes are there when 1,1-dimethylcycloheptane…
A: In presence of light alkanes and cycloalkanes give halogenation reaction.This reaction proceeds…
Q: Identify the major product in the following reaction sequence. Br₂ / light DBN NBS HBr peroxide…
A:
Q: 2. Give the resonance structures for the species given below. HO NH Which atom is more basic in the…
A: A question based on introduction to organic chemistry. Based on the tautomerism concept the given…
Q: The reaction N2O4↽−−⇀2NO2 is allowed to reach equilibrium in a chloroform solution at 25 ∘C .…
A:
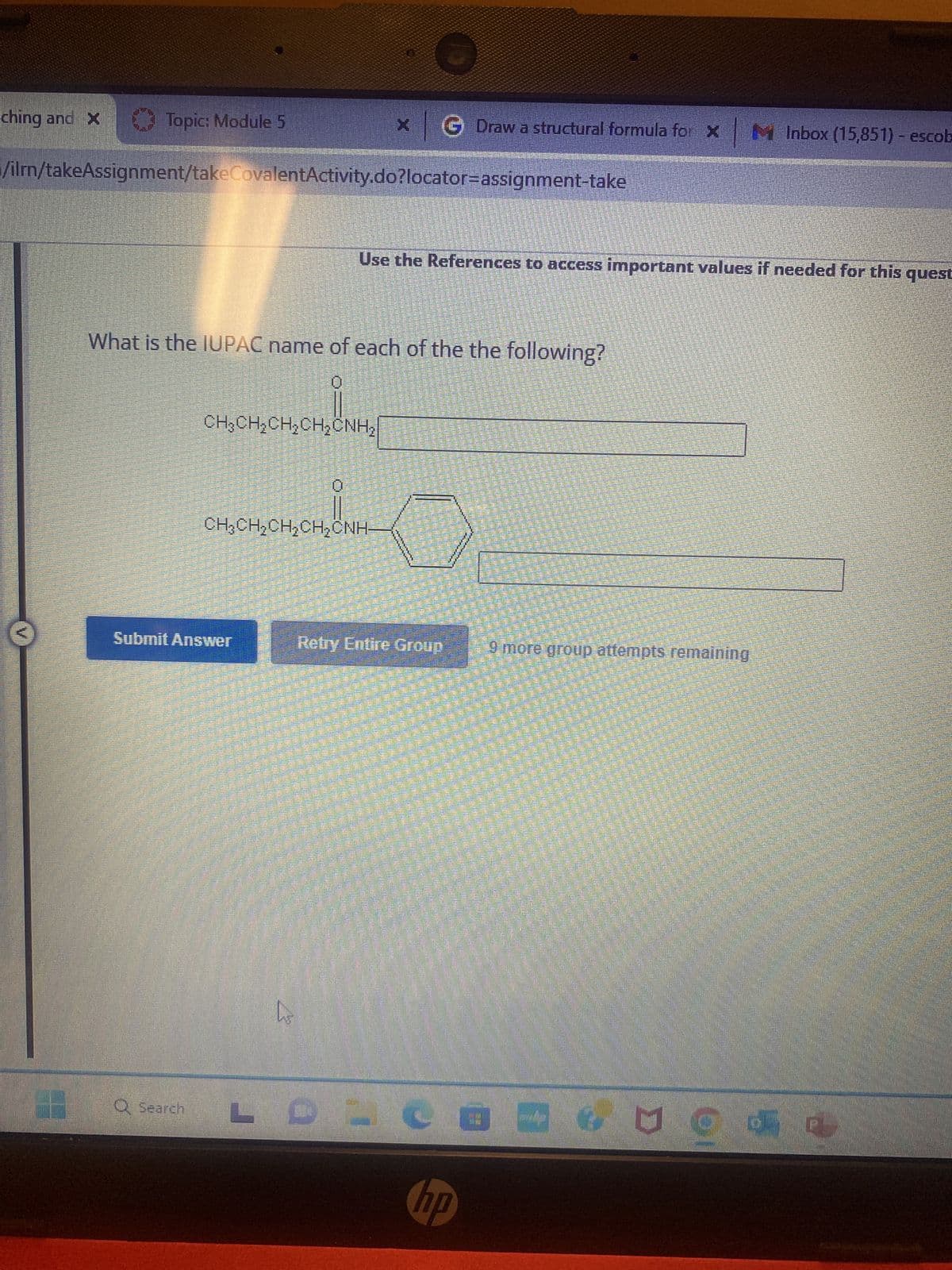
Q: Draw a structural formula for pentanoic acid. • You do not have to consider stereochemistry. • You…
A: A formula which shows the arrangement of atoms in the molecule of compound is called structural…
Q: Case Study 4 Salicylic Acid Ointment 2% You receive a new prescription from an older gentleman…
A: we'll start by calculating the amount of salicylic acid and soft white paraffin…
Q: Using the balanced equation: 2C₂H6S(g) +902(g) → 4CO2(g) +2SO₂(g) + 6H₂O(g) and the diagram below,…
A: From the given diagram:The number of units of C2H6S = 3The number of units of O2 = 9
Q: Macmillan Learning Consider the given reaction. おえ一枚 Which reactant is the Lewis acid and which is…
A: It is based on the concept of Lewis acid and Base.Here we are required to identify the Lewis acid…
Q: Select the correct IUPAC name for the cycloalkane: F. Br The IUPAC name is:…
A: According to IUPAC Nomenclature,Name of alicyclic compounds are obtained by adding prefix cyclo to…
Q: Select the molecules that are polar. □ IOF 5 AsF3 CF4 CH₂Cl₂
A: A question based on molecules. 4 molecules are given that are to be distinguished based on their…
Q: A student separates colored compounds A and B (shown in grey in the figure below) using column…
A: A question based on analytical process. Based on the Column chromatography and the polarity index,…
Q: Pick the IUPAC name of the moelucle shown below. H H₂CO H O 4-methoxy-2.3 epoxyhexane O2.3…
A: The IUPAC is used to determine the name of the compound based on different functional group present…
Q: Aspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3) with salicylic acid…
A: The given balanced reaction of formation of aspirin by the reaction of acetic anhydride and…
Q: Part D. Do the two structures A and B of each pair drawn below represent the same molecule,…
A: Isomers: Same molecular formula but different structure. Constitutional Isomers: Same molecular…
Q: Draw the major organic product for the reaction. H H₂O cat. TfOH Select Draw Templates More C H O E…
A: Hydration of alkynes in TfOH (trifluoromethanesulfonic acid) is a chemical reaction that involves…
Q: 18000 10000 14000- 12000 10000 8 8000. 6000- 4000- 2000- 0 0 0.5 Component Acetone 0.82 Isoborneol…
A: Retention time is defined as the time that a solute spends in a column or the time spent in the…
Q: Question 10 Gas chromatography is: a recording of velocity versus time the flash point values versus…
A: A question based on analytical process. 5 choices about the gas chromatography is given the most…
Q: 9. Give two names for each of the following: CH3CH₂ H₂C a. CH3CH2 C-CHCHCH3 CH3 b. CH3 C-CHCH₂CH3
A: Here we can see that we have an epoxide group as a…
Q: What volume of 0.181 MNa3PO4 solution is necessary to completely react with 87.9 mL of 0.105 MCuCl2
A: The balanced reaction between Na3PO4 and CuCl2 is
Q: (1) (a) and (b) are elements that belong to period 5 and 6 in the periodic table, respectively. Both…
A: (1)The position of silver (Ag) is in the 5th period of the periodic table and lead (Pb) is in the…
Q: The pesticide DDT, C14H9Cl5 is produced by the following reaction 2C6H5Cl + C2HOCl3 - C14H9Cl5 + H2O…
A: Amount of C2HOCl3 = 34.5 gAmount of C6H5Cl = 22.8 gMolar mass of C6H5Cl = 112.56 g/molMolar mass of…
Q: 2. Is the molecule shown below chiral or achiral? CH2OH HO₂C H CO₂H
A:
Q: нн H Н НН Н н
A: An ether molecule
Q: 2 molecules of A come together to form an AA dimer. The rate of binding is slightly less than…
A: Dimerization of molecule ARate of forward reaction, i.e Binding or dimerization A-A= 2X106…
Q: 2. To save on production costs, r 200 g of chromium. In order t standard bumper should be imm [1.48…
A: Time:The time can be evaluated by the relationship between the charge and the current produced by…
Q: The pH of a 1.3M solution of alloxanic acid (HC₂H₂N₂O) is measured to be 3.27. Calculate the acid…
A:
Q: Pa is 96% theol air at Liquid octane at temperature is 600K. How much energy is emitted to the…
A:
Q: Consider the three-dimensional structure shown. Note that only the bonding groups (outer atoms) are…
A: Bond pairslone payersBond angleshape of the molecule10not…
Q: A balloon filled with helium has a volume of 7.0 L. What is the mass, in grams, of helium in the…
A: Given:Volume of helium gas = 7.0 LMolar mass of helium gas = 4 g mol-1
Q: Which of the molecules are linear? BeCl₂ SF 2 NO₂ XeF2
A:
Q: Identify the axial and equatorial atoms in the three-dimensional structure. What is the shape of…
A:
Q: How many structures are possible for a trigonal bipyramidal molecule with a formula of AX₂ Y₂?…
A: Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It…
Q: For the gas phase decomposition of hydrogen iodide at 700 K 2 HI-H₂+1₂ updates now or try ton the…
A:
Q: Which of the following best describes the experimental conditions under which an alkane can be…
A: Halogenation of alkanes such as Chlorination and Bromination takes place via free radical mechanism…
Q: Iron is one of our most important metals since it is cheap and very abundant. Iron can also be mixed…
A: In the given reaction:FeO + CO -> Fe + CO2The reducing agent is the species that donates…
Q: NEED presentation any topic u own choose but topic is good and with reactions and awesome ok i have…
A: Let's proceed step by step. For the topic of your presentation, I suggest "Nuclear Chemistry and its…
Q: Consider the following equilibrium: Hind + H₂O H3O+¹ + Ind-¹ At the transition point for an…
A: At transition point, solution changes colour. At transition point, [HIn] = [In-] HIn = indicator ,…
Q: Ⓒ Macmillan Learning Identify the molecular shape of each of the molecules. Molecule SICI SF *+…
A: The arrangement of atoms in a molecule in three dimensions is known as molecular geometry, commonly…
Q: Use the data given in the table below to draw the S(T)-diagram for the heating up of ethanol from…
A: The table shows the values of Cp and enthalpy of phase changePhase TransitionsCp/J mol-1K-1s(Heating…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 4 images

- A solution of altura red of concentration 307 ppm gives an absorbance of 0.616. What concentration is a solution of allura red which gives an absorbance reading of 0.261? The same cell (cuvette) with a path length of 1.000 cm is used for both readings. Report your answer to the nearest whole number without units? Hint: A=abc, where c can have any concentration units. dont provide handwriten solutionGive only typing answer with explanation and conclusion A solution of Tryptophan has an absorbance at 280nm of 0.54 in a 0.5 cm length cuvette. Given the absorbance coefficient of trp 6.4 x 103 L Mol-1 cm-1. What is the concentration of solution? Please indicate the formula clearly.a) Draw tge HY^3- species of EDTA to illustrate that it is a Zwitterion. b) Explain the function of a SEM by drawing a sketch and giving caption for it.
- You are tasked with creating a simplified pourboiux diagram of silicia speciation. The relevent reactions are HSO4- <-> SO42- , H2S <-> HS-, and S <-> H2S. The delta G of S is 0, HSO4- = -755.3, SO42- -744.0, H2S = -27.7, HS- = 12.2 The silica activity is 10^-3 M. Set and solve the reactions so they can be graphed on a pe/pH diagram.As part of an investigation on Pb in biological ecosystems, some grasses that grew along the road and were exposed to emissions from motor gasoline were collected. 6.2501 g of grass were calcined to destroy all organic matter, the inorganic residue was properly treated and finally made up to 100 mL. An aliquot of 50 mL of this solution was read in an atomic absorption spectrophotometer registering an absorbance of 0.1250; then 0.10 mL of a standard solution of 55 µg of Pb / mL were added to the other 50 mL, with this fortified sample, another absorbance measurement was made, which was 0.1798. Calculate the µg of Pb / g of grassA 0.1017-g sample of KBrO3 (MW = 167 g/mole) was dissolved in dilute HCl and treated with an unmeasured excess of KI. The liberated iodine required 39.75 mL of a sodium thiosulfate solution. Calculate the molar concentration of the Na2S2O3. BrO3 - + 6I + 6HCl → Br - + 3I2 + 6KCl + 3H2O I2 + Na2S2O3 → 2NaI + Na2S4O6 The type of redox titration reaction is ___. a. iodimetry b. bromination c. iodometry d. diazotination The role of KBrO3 in the titration is ___. a. reducing agent b. oxidizing agent c. source of Br2 d. source of I2 At the endpoint of titration, the equivalence of sodium thiosulfate is equal to the following, EXCEPT ___ . a. equivalence of KI b. equivalence of liberated I2 c. equivalence of KBrO3 d. none of the other choices
- A cobalt(II) salt was dissolved in water and reacted with excess acetylacetone (1,2-pentanedione, CH3 COCHCOCH3 ) and hydrogen peroxide. A green solid was formed that gave the following results for elemental analysis: C, 50.4 per cent; H, 6.2 per cent; Co, 16.5 per cent (all by mass). Determine the ratio of cobalt to acetylacetonate ion in the product.A newly synthesized drug was found to have a carboxyl group, an imide and a sulfonamide group. The first type of method to consider in this drug molecule’s quantitatve analysis is: a. Non-aqueous Alkalimetryb. Non-aqueous Acidimetryc. Aqueous Alklaimetryd. Aqueous AcidimetryA 0.9056 g sample of KBrO3 (MM=167.0) was dissolved in dilute HCl and treated with an unmeasured excess of KI. The liberated iodine required 37.21 mL of Na2S2O3. Calculate the molar concentration of sodium thiosulfate. 1 mol BrO3- = 3 mol I2 = 6 mol S2O32- 0.8744 M 2.623 M 0.8791 M 0.2915 M
- You and a partner are asked to complete a lab entitled“Oxides of Ruthenium” that is scheduled to extend over twolab periods. The first lab, which is to be completed by yourpartner, is devoted to carrying out compositional analysis.In the second lab, you are to determine melting points. Upongoing to lab you find two unlabeled vials, one containinga soft yellow substance and the other a black powder. Youalso find the following notes in your partner’s notebook—Compound 1: 76.0% Ru and 24.0% O (by mass), Compound2: 61.2% Ru and 38.8% O (by mass). (a) What is the empiricalformula for Compound 1? (b) What is the empiricalformula for Compound 2? Upon determining the meltingpoints of these two compounds, you find that the yellowcompound melts at 25 °C, while the black powder does notmelt up to the maximum temperature of your apparatus,1200 °C. (c) What is the identity of the yellow compound?(d) What is the identity of the black compound? (e) Whichcompound is molecular? (f) Which compound is…I need backgorund information of ferrocene and it's components also who found it. Thank you so much!A phenol standard with a concentration of 4.00 ppm has an absorbance of 0.424 at a wavelength of 460 nm using a 1.00-cm cell. A water sample is steam-distilled, and a 50.00-mL aliquot of the distillate is placed in a 100-mL volumetric flask and diluted to volume with distilled water. The absorbance of this solution is found to be 0.394. What is the concentration of phenol (in parts per million) in the water sample?

