Q: Consider the insoluble compound copper(II) hydroxide, Cu(OH)2. The copper(II) ion also forms a…
A:
Q: create a net ionic equation for the reaction between the following aqueous solutions: a)(CH3)3NHBr…
A: Answer:- This question is answered by using the simple concept of writing the net ionic equation in…
Q: A compound with molecular formula C6H10 displays the following IR and Mass Spectrum. a) Calculate…
A: The IR spectrum and mass spectrum of a compound having a molecular formula of C6H10 have been…
Q: To answer the questions, interpret the following Lewis diagram for NO₂F. :O-N=0 | F: 1. For the…
A:
Q: What is the crystal structure for An2Da? Neatly draw its unit cell, using proper notation.
A: As per the student's request I am solving only part-C .
Q: What will be the appropriate reagents in the following reactions? reagent A reagent B reagent C Br…
A: Given reaction is multi step synthesis. In first three reactions halogenation take place. An last…
Q: TO b) A A CI B B NH3 CC13 C OMe most reactive most reactive least reactive least reactive
A:
Q: Draw the Lewis structure for SF5CI in the window below and then answer the questions that follow. O…
A: Lewis Structure is a representation of the valence shell electrons in a molecule. It is used to show…
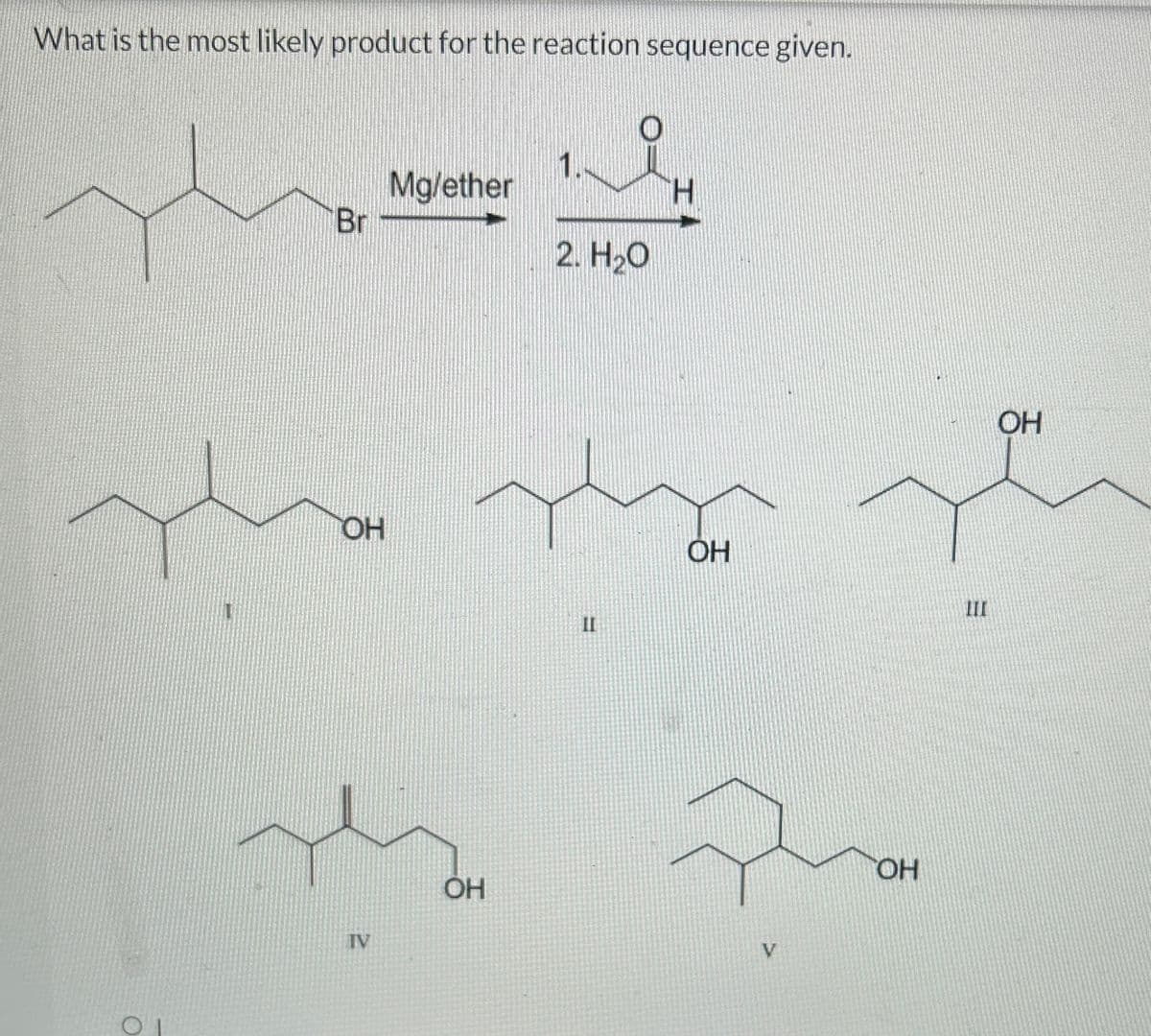
Q: 3. Provide the product(s) or reagent(s) as appropriate. CI ? 1. H₂O, H₂SO4 2. H₂CrO4 1. NaOH 2.…
A: We have to give the reagents and products of the given reactions. Introduction:…
Q: A student sets up the following equation to convert a measurement. (The ? stands for a number the…
A: Here we are required to find the conversion factors which is required by the student to covert the…
Q: What are possible targets of anticancer therapies? Olipids enzymes Onucleic acids Oglycans
A: Numerous medications used to treat cancer use various mechanisms of action. Targeting drugs at…
Q: When chemical A and chemical B are mixed together, and endothermic reaction below occurs:…
A: Firat we need to calculate the heat of reaction. Then we will calculate moles of each reactant and…
Q: Identify the geometry and bond angles for the following unknown 3 compound: XF4, where X is an…
A: The first compound may be Sef4 And second may be BrF5
Q: The following compound is chiral Oachiral Co C=C=C «Η CH3 will
A: Chiral compounds are those compounds which have chiral center with no plane of symmetry and some…
Q: Counting each stereoisomer separately, how many different products can be generated from the…
A: Option 3 is correct answer for the given question because total number of Stereoisomers are 4 .
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A: In this question, we will draw the structure of the major organic product. In this reaction, HBr is…
Q: What mass precipitate will form if 0.300 L of 0.190 M Nal is mixed with excess Pb(ClO3)2?…
A:
Q: can 1,2-dibromobenzene and 1,3-dibromobenzene distinguishe by H NMR spectroscopy? describe number of…
A:
Q: fo H₂C-00 H₂C-0 -(CH₂) 14CH3 (CH₂)C=C(CH₂),CH, H H -(CH₂) 16CH3 3NaOH, H₂O H₂C-OH HC-OH H₂C-OH +
A:
Q: Predict the reagent(s) needed to produce this regiochemistry in this elimination reaction. Br Q
A: Organic reactions are those in which organic reactant react to form organic product. In the given…
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A: we have to calculate the percentage yield of water in the given reaction
Q: If 5.50 L of water vapor at 50.2 °C and 0.121 atm reacts with excess iron, how many grams of…
A: Recall the given reaction,♣ 2 Fe s + 3 H2O g → Fe2O3 s + 3…
Q: 1. Ascorbic acid (Vitamin C) reacts with 13 using starch as the indicator. The endpoint is marked by…
A: Vitamin C is ascorbic acid (C6H8O6) and it reacts with triiodide ion (I3-) a shown by the following…
Q: Kinetics study on the decomposition of phosphine at 950 K was done by injecting PH3 gas into a…
A: A question based on chemical kinetics. Two sub parts are given based on a set of kinetic data, for…
Q: A solution of a weak acid, HA, has [HA] = [A] = 0.271 M. If the acid dissociation constant for the…
A:
Q: Assume that a growth curve was done at a less-than-optimal salt concentration. Predict how the…
A: Salts like NaCl, KCl, Na2SO4 etc are the required nutrients that effect the growth of different…
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Answer: Entropy change is the measure of system's disorderliness. In more possible states, system…
Q: Calculate the concentration of metal ions, M³+, in a solution that initially contains 0.20 M M(NO3)3…
A: Formation reaction [M(CN)6]3- M3+ + 6CN- <--> [M(CN)6]3-
Q: 11 [Ca], equilibrium (mol/L) 12 Molar solubility of Ca(OH), (mol/L) Average molar solubility of…
A: Saturated Calcium hydroxide in water is in equilibrium with Ca2+ and OH- according to the…
Q: A wooden artifact from a Chinese temple has a ¹4C activity of 24.9 counts per minute as compared…
A: we have to calculate the age of the sample of wooden artifact
Q: Design a buffer that has a pH of 10.00 using one of the weak base/conjugate acid systems shown…
A: According to the question, The pH of the buffer is given by = 10.00 For the pH 10, The buffer lies…
Q: What term is used for each quantity? (For example, 0.001 g=one milligram) A. 0.000 001g B.…
A: Answer:- Given A. 0.000001 g B. 1000000000 m C. 0.000000001 s D. 0.01 g What term is used for…
Q: What mass of water is produced from the complete combustion of 3.70×10−3 g of methane?
A: Given, mass of methane (CH4) react = 3.70 × 10-3 g mass of water (H2O) produced = ? Note: molar mass…
Q: Be sure to answer all parts. An FM station broadcasts classical music at 104.3 MHz (megahertz, or…
A:
Q: Calculate the volume percent of solute in each of the solutions. A solution made by adding 20.3 mL…
A: The volume percent of solute in a solution is given by the equation, Volume percent (%v/v) =Volume…
Q: 1.172kg of an unknown gas comprised of only carbon and hydrogen is held in a 50.0L vessel at 25.0°C.…
A: Mass of Unknown gas = 1.172 kg = 1.172 x 10 3 g Volume (V) = 50.0 L Temperature (T) = 25.0°C =298.15…
Q: When ammonia (NH3) is used as a solvent, the leveling effect will prevent the presence of bases…
A: All bases tend to become indistinguishable in strength when dissolved in Ammonia owing to the…
Q: Question 2 of 7 View Policies Show Attempt History Current Attempt in Progress * Incorrect. 0/1 E…
A: The term "unsaturated" is used when the carbon atoms in a fat molecule do not have the maximum…
Q: A solution contains 1.47x10-2 M magnesium acetate and 1.07x10-2 M manganese(II) nitrate. Solid…
A: According to the question, The concentration of the magnesium acetate is given = 1.47×10-2M The…
Q: For each of the metabolic transformations (e) through (h), determine whether the compound on the…
A:
Q: Assuming an efficiency of 44.30%, calculate the actual yield of magnesium nitrate formed from 122.9…
A: percent yield = actual yield / theoretical yield x 100%.
Q: Use the phase diagram of Substance x below to find the pressure at which the boiling point of…
A: Boiling point: The temperature at which the vapor pressure of the liquid becomes equal to the…
Q: I am trying to find the mass of my compound, but Im not sure if its 76 or 173 bc Im not sure if…
A: To determine the molecular ion peak in the given mass spectra.
Q: Isopropyl alcohol is mixed with water to produce a 34.0% (v/v) alcohol solution. How many…
A: The volume percent of isopropyl alcohol = 34.0 % (v/v) Total volume of the solution = 735 mL
Q: 2. When you mix two solutions one containing compound A and the other containing compound B in a…
A: Because the final temperature for the mixing of compound A and compound B is increase tgen it is a…
Q: At constant temperature, the pressure on a 6.0 L sample of gas is increased from 1.0 to 2.0 atm.…
A: Boyle's law This law states that the absolute pressure exerted by a given mass of an ideal gas is…
Q: How much heat is necessary to melt a block of ice that is 2.75 cm X 6.48 cm X 17.32 cm? (Density of…
A: When an ice block melted it absorbs heat and the required heat at a constant temperature is known as…
Q: From data found in appendix G, calculate the enthalpy of the following reaction: 2 PbO(s, yellow) +…
A:
Q: What would be the best sequence of reactions/reagents to use for this transformation (the one that…
A: We will use reagents given and do given organic conversion.
Q: Draw the major product of this E1 reaction. Ignore any inorganic byproducts.
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Explain why Jone's oxidation of 4-methyl-2-pentanol is faster than that of 3,3-dimethyl-2-pentanol.Why is this considered an Elimination E2 reaction? C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol → propene + waterExplain why the trifluoromethane sulfonate (TfO-) ion is a better leaving group than the methanesulfonate (MsO-) ion.
- The product of reaction between acetone and bromine (excess) with KOH excess is A. Ch2BrCOCh3 B. ChBr3 C.ChBr2COCH3 D.CBr3COCH3 E.Ch2BrCOCHBr2Show how to make these deuterium-labeled compounds, using CD3MgBr and D2O as your sources of deuterium, and anynon-deuterated starting materials you wish.(a) CH3CH(OD)CD3Alkyl diazonium salts (shown below) are considered "super" leaving groups; a consequence of this is that they tend to be contact explosives What quailities make alkyl diazonium salts such excellent leaving groups?
- Show the SN2 mechanism in the reaction between (S) 2-bromobutane and iodide ion. Label the product and draw a enery diagram for this reaction.Which halide in the attached marine natural product reacts fastest in the SN1 reaction?Which compound in each of the following pairs will react faster in SN2 reaction with —OH?(i) CH3Br or CH3I (ii) (CH3)3 CCl or CH3Cl
- Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.Which halide in the attached marine natural product reacts fastest inthe SN2 reaction?The name for Reaction 1 is _____ while Reaction 2 is called _____. Choices: A. Williamson Ether synthesis, B. Hydration, C. Epoxidation, D. Acidic cleavage The reagent/s for REACTION 1 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. H2O/H3PO4, C. H3O+, D. HI The reagent/s for REACTION 2 is/are ______ Choices: A. m-chloroperoxybenzoic acid, B. water in acidic medium, C. dilute acid, D. hydroiodic acid

